Extracellular matrix assembly: a multiscale deconstruction
- PMID: 25370693
- PMCID: PMC4682873
- DOI: 10.1038/nrm3902
Extracellular matrix assembly: a multiscale deconstruction
Abstract
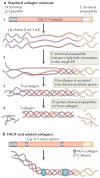
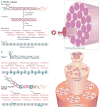
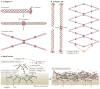
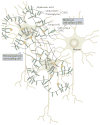
The biochemical and biophysical properties of the extracellular matrix (ECM) dictate tissue-specific cell behaviour. The molecules that are associated with the ECM of each tissue, including collagens, proteoglycans, laminins and fibronectin, and the manner in which they are assembled determine the structure and the organization of the resultant ECM. The product is a specific ECM signature that is comprised of unique compositional and topographical features that both reflect and facilitate the functional requirements of the tissue.
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Targeted proteomics effectively quantifies differences between native lung and detergent-decellularized lung extracellular matrices.Acta Biomater. 2016 Dec;46:91-100. doi: 10.1016/j.actbio.2016.09.043. Epub 2016 Sep 29. Acta Biomater. 2016. PMID: 27693690 Free PMC article.
-
The bi-functional organization of human basement membranes.PLoS One. 2013 Jul 3;8(7):e67660. doi: 10.1371/journal.pone.0067660. Print 2013. PLoS One. 2013. PMID: 23844050 Free PMC article.
-
Mechanical properties of basement membrane in health and disease.Matrix Biol. 2017 Jan;57-58:366-373. doi: 10.1016/j.matbio.2016.07.001. Epub 2016 Jul 17. Matrix Biol. 2017. PMID: 27435904 Review.
-
Extracellular matrix components in the pathogenesis of type 1 diabetes.Curr Diab Rep. 2014 Dec;14(12):552. doi: 10.1007/s11892-014-0552-7. Curr Diab Rep. 2014. PMID: 25344787 Free PMC article. Review.
-
[Structural elements and conformation of basement membranes].Postepy Hig Med Dosw. 2000;54(1):83-97. Postepy Hig Med Dosw. 2000. PMID: 10803026 Review. Polish.
Cited by
-
Design of Bio-Conjugated Hydrogels for Regenerative Medicine Applications: From Polymer Scaffold to Biomolecule Choice.Molecules. 2020 Sep 7;25(18):4090. doi: 10.3390/molecules25184090. Molecules. 2020. PMID: 32906772 Free PMC article. Review.
-
ADAMTS9-Mediated Extracellular Matrix Dynamics Regulates Umbilical Cord Vascular Smooth Muscle Differentiation and Rotation.Cell Rep. 2015 Jun 16;11(10):1519-28. doi: 10.1016/j.celrep.2015.05.005. Epub 2015 May 28. Cell Rep. 2015. PMID: 26027930 Free PMC article.
-
Formation of pre-metastatic bone niche in prostate cancer and regulation of traditional chinese medicine.Front Pharmacol. 2022 Aug 17;13:897942. doi: 10.3389/fphar.2022.897942. eCollection 2022. Front Pharmacol. 2022. PMID: 36059977 Free PMC article. Review.
-
Modulating barriers of tumor microenvironment through nanocarrier systems for improved cancer immunotherapy: a review of current status and future perspective.Drug Deliv. 2020 Dec;27(1):1248-1262. doi: 10.1080/10717544.2020.1809559. Drug Deliv. 2020. PMID: 32865029 Free PMC article. Review.
-
Decellularized extracellular matrix-based disease models for drug screening.Mater Today Bio. 2024 Sep 27;29:101280. doi: 10.1016/j.mtbio.2024.101280. eCollection 2024 Dec. Mater Today Bio. 2024. PMID: 39399243 Free PMC article. Review.
References
-
- Mecham RP. Overview of extracellular matrix. Curr Protoc Cell Biol. 2012;10(Unit 10.1) - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

