Human RecQ helicases in DNA repair, recombination, and replication
- PMID: 24606147
- PMCID: PMC4586249
- DOI: 10.1146/annurev-biochem-060713-035428
Human RecQ helicases in DNA repair, recombination, and replication
Abstract
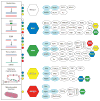
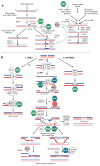
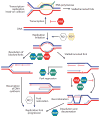
RecQ helicases are an important family of genome surveillance proteins conserved from bacteria to humans. Each of the five human RecQ helicases plays critical roles in genome maintenance and stability, and the RecQ protein family members are often referred to as guardians of the genome. The importance of these proteins in cellular homeostasis is underscored by the fact that defects in BLM, WRN, and RECQL4 are linked to distinct heritable human disease syndromes. Each human RecQ helicase has a unique set of protein-interacting partners, and these interactions dictate its specialized functions in genome maintenance, including DNA repair, recombination, replication, and transcription. Human RecQ helicases also interact with each other, and these interactions have significant impact on enzyme function. Future research goals in this field include a better understanding of the division of labor among the human RecQ helicases and learning how human RecQ helicases collaborate and cooperate to enhance genome stability.
Keywords: BLM; RECQL1; RECQL4; RECQL5; WRN; genome stability.
Figures

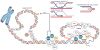
Similar articles
-
Roles of RECQ helicases in recombination based DNA repair, genomic stability and aging.Biogerontology. 2009 Jun;10(3):235-52. doi: 10.1007/s10522-008-9205-z. Epub 2008 Dec 15. Biogerontology. 2009. PMID: 19083132 Free PMC article. Review.
-
Rising from the RecQ-age: the role of human RecQ helicases in genome maintenance.Trends Biochem Sci. 2008 Dec;33(12):609-20. doi: 10.1016/j.tibs.2008.09.003. Epub 2008 Oct 14. Trends Biochem Sci. 2008. PMID: 18926708 Free PMC article. Review.
-
Roles of the Werner syndrome RecQ helicase in DNA replication.DNA Repair (Amst). 2008 Nov 1;7(11):1776-86. doi: 10.1016/j.dnarep.2008.07.017. Epub 2008 Sep 6. DNA Repair (Amst). 2008. PMID: 18722555 Free PMC article. Review.
-
Human RecQ Helicases in DNA Double-Strand Break Repair.Front Cell Dev Biol. 2021 Feb 25;9:640755. doi: 10.3389/fcell.2021.640755. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 33718381 Free PMC article. Review.
-
Human RECQL5: guarding the crossroads of DNA replication and transcription and providing backup capability.Crit Rev Biochem Mol Biol. 2013 May-Jun;48(3):289-99. doi: 10.3109/10409238.2013.792770. Epub 2013 Apr 29. Crit Rev Biochem Mol Biol. 2013. PMID: 23627586 Free PMC article. Review.
Cited by
-
Using a Function-First "Scout Fragment"-Based Approach to Develop Allosteric Covalent Inhibitors of Conformationally Dynamic Helicase Mechanoenzymes.J Am Chem Soc. 2024 Jan 10;146(1):62-67. doi: 10.1021/jacs.3c10581. Epub 2023 Dec 22. J Am Chem Soc. 2024. PMID: 38134034 Free PMC article.
-
DNA damage processing at telomeres: The ends justify the means.DNA Repair (Amst). 2016 Aug;44:159-168. doi: 10.1016/j.dnarep.2016.05.022. Epub 2016 May 16. DNA Repair (Amst). 2016. PMID: 27233113 Free PMC article. Review.
-
Recognition and unfolding of human telomeric G-quadruplex by short peptide binding identified from the HRDC domain of BLM helicase.RSC Adv. 2022 Aug 8;12(34):21760-21769. doi: 10.1039/d2ra03646k. eCollection 2022 Aug 4. RSC Adv. 2022. PMID: 36043100 Free PMC article.
-
Telomere organization and the interstitial telomeric sites involvement in insects and vertebrates chromosome evolution.Genet Mol Biol. 2022 Nov 14;45(3 Suppl 1):e20220071. doi: 10.1590/1678-4685-GMB-2022-0071. eCollection 2022. Genet Mol Biol. 2022. PMID: 36394537 Free PMC article.
-
WRN Mutation Update: Mutation Spectrum, Patient Registries, and Translational Prospects.Hum Mutat. 2017 Jan;38(1):7-15. doi: 10.1002/humu.23128. Epub 2016 Oct 7. Hum Mutat. 2017. PMID: 27667302 Free PMC article. Review.
References
-
- Larsen NB, Hickson ID. RecQ helicases: conserved guardians of genomic integrity. Adv Exp Med Biol. 2013;767:161–84. - PubMed
-
- Mandell JG, Goodrich KJ, Bahler J, Cech TR. Expression of a RecQ helicase homolog affects progression through crisis in fission yeast lacking telomerase. J Biol Chem. 2005;280:5249–57. - PubMed
-
- Barea F, Tessaro S, Bonatto D. In silico analyses of a new group of fungal and plant RecQ4-homologous proteins. Comput Biol Chem. 2008;32:349–58. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

