Targeted mutagenesis of mitochondrial carbonic anhydrases VA and VB implicates both enzymes in ammonia detoxification and glucose metabolism
- PMID: 23589845
- PMCID: PMC3645511
- DOI: 10.1073/pnas.1305805110
Targeted mutagenesis of mitochondrial carbonic anhydrases VA and VB implicates both enzymes in ammonia detoxification and glucose metabolism
Abstract
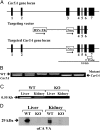
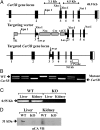
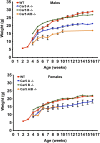
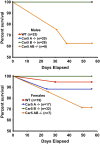
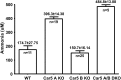
Prior studies with carbonic anhydrase (CA) inhibitors implicated mitochondrial CA in ureagenesis and gluconeogenesis. Subsequent studies identified two mitochondrial CAs. To distinguish the contribution of each enzyme, we studied the effects of targeted disruption of the murine CA genes, called Car5A and Car5B. The Car5A mutation had several deleterious consequences. Car5A null mice were smaller than wild-type littermates and bred poorly. However, on sodium-potassium citrate-supplemented water, they produced offspring in expected numbers. Their blood ammonia concentrations were markedly elevated, but their fasting blood sugars were normal. By contrast, Car5B null mice showed normal growth and normal blood ammonia levels. They too had normal fasting blood sugars. Car5A/B double-knockout (DKO) mice showed additional abnormalities. Impaired growth was more severe than for Car5A null mice. Hyperammonemia was even greater as well. Although fertile, DKO animals were produced in less-than-predicted numbers even when supplemented with sodium-potassium citrate in their drinking water. Survival after weaning was also reduced, especially for males. In addition, fasting blood glucose levels for DKO mice were significantly lower than for controls (153 ± 33 vs. 230 ± 24 mg/dL). The enhanced hyperammonemia and lower fasting blood sugar, which are both seen in the DKO mice, indicate that both Car5A and Car5B contribute to both ammonia detoxification (ureagenesis) and regulation of fasting blood sugar (gluconeogenesis). Car5A, which is expressed mainly in liver, clearly has the predominant role in ammonia detoxification. The contribution of Car5B to ureagenesis and gluconeogenesis was evident only on a Car5A null background.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Mitochondrial carbonic anhydrase VA and VB: properties and roles in health and disease.J Physiol. 2023 Jan;601(2):257-274. doi: 10.1113/JP283579. Epub 2022 Dec 19. J Physiol. 2023. PMID: 36464834 Free PMC article. Review.
-
Regulation of high glucose-induced apoptosis of brain pericytes by mitochondrial CA VA: A specific target for prevention of diabetic cerebrovascular pathology.Biochim Biophys Acta Mol Basis Dis. 2017 Apr;1863(4):929-935. doi: 10.1016/j.bbadis.2017.01.025. Epub 2017 Jan 26. Biochim Biophys Acta Mol Basis Dis. 2017. PMID: 28131914 Free PMC article.
-
Carbonic anhydrase IV and XIV knockout mice: roles of the respective carbonic anhydrases in buffering the extracellular space in brain.Proc Natl Acad Sci U S A. 2005 Nov 15;102(46):16771-6. doi: 10.1073/pnas.0508449102. Epub 2005 Oct 31. Proc Natl Acad Sci U S A. 2005. PMID: 16260723 Free PMC article.
-
Ammonia detoxification via ureagenesis in rat hepatocytes involves mitochondrial aquaporin-8 channels.Hepatology. 2013 May;57(5):2061-71. doi: 10.1002/hep.26236. Hepatology. 2013. PMID: 23299935
-
Comprehensive characterization of ureagenesis in the spfash mouse, a model of human ornithine transcarbamylase deficiency, reveals age-dependency of ammonia detoxification.J Inherit Metab Dis. 2019 Nov;42(6):1064-1076. doi: 10.1002/jimd.12068. Epub 2019 Mar 13. J Inherit Metab Dis. 2019. PMID: 30714172 Review.
Cited by
-
Mitochondrial carbonic anhydrase VA and VB: properties and roles in health and disease.J Physiol. 2023 Jan;601(2):257-274. doi: 10.1113/JP283579. Epub 2022 Dec 19. J Physiol. 2023. PMID: 36464834 Free PMC article. Review.
-
Transgenic expression of carbonic anhydrase III in cardiac muscle demonstrates a mechanism to tolerate acidosis.Am J Physiol Cell Physiol. 2019 Nov 1;317(5):C922-C931. doi: 10.1152/ajpcell.00130.2019. Epub 2019 Aug 7. Am J Physiol Cell Physiol. 2019. PMID: 31390226 Free PMC article.
-
Carbonic Anhydrases: Role in pH Control and Cancer.Metabolites. 2018 Feb 28;8(1):19. doi: 10.3390/metabo8010019. Metabolites. 2018. PMID: 29495652 Free PMC article. Review.
-
Carbonic anhydrase III (Car3) is not required for fatty acid synthesis and does not protect against high-fat diet induced obesity in mice.PLoS One. 2017 Apr 24;12(4):e0176502. doi: 10.1371/journal.pone.0176502. eCollection 2017. PLoS One. 2017. PMID: 28437447 Free PMC article.
-
Characterizing the metabolic divide: distinctive metabolites differentiating CAD-T2DM from CAD patients.Cardiovasc Diabetol. 2024 Jan 6;23(1):14. doi: 10.1186/s12933-023-02102-0. Cardiovasc Diabetol. 2024. PMID: 38184583 Free PMC article.
References
-
- Lindskog S, Silverman DN. In: The Carbonic Anhydrases: New Horizons. Chegwidden WR, Carter ND, Edwards YH, editors. Basel, Switzerland: Birkhauser; 2000. pp. 175–195.
-
- Sly WS, Hu PY. Human carbonic anhydrases and carbonic anhydrase deficiencies. Annu Rev Biochem. 1995;64:375–401. - PubMed
-
- Supuran CT. Carbonic anhydrases: Novel therapeutic applications for inhibitors and activators. Nat Rev Drug Discov. 2008;7(2):168–181. - PubMed
-
- Hewett-Emmett D. In: The Carbonic Anhydrases: New Horizons. Chegwidden WR, Carter ND, Edwards YH, editors. Basel, Switzerland: Birkhauser; 2000. pp. 29–76.
-
- Lehtonen J, et al. Characterization of CA XIII, a novel member of the carbonic anhydrase isozyme family. J Biol Chem. 2004;279(4):2719–2727. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

