Latent TGF-β binding protein 4 promotes elastic fiber assembly by interacting with fibulin-5
- PMID: 23382201
- PMCID: PMC3581912
- DOI: 10.1073/pnas.1215779110
Latent TGF-β binding protein 4 promotes elastic fiber assembly by interacting with fibulin-5
Abstract
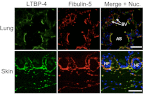
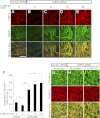
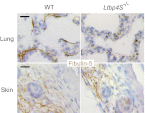
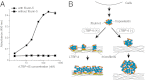
Elastic fiber assembly requires deposition of elastin monomers onto microfibrils, the mechanism of which is incompletely understood. Here we show that latent TGF-β binding protein 4 (LTBP-4) potentiates formation of elastic fibers through interacting with fibulin-5, a tropoelastin-binding protein necessary for elastogenesis. Decreased expression of LTBP-4 in human dermal fibroblast cells by siRNA treatment abolished the linear deposition of fibulin-5 and tropoelastin on microfibrils. It is notable that the addition of recombinant LTBP-4 to cell culture medium promoted elastin deposition on microfibrils without changing the expression of elastic fiber components. This elastogenic property of LTBP-4 is independent of bound TGF-β because TGF-β-free recombinant LTBP-4 was as potent an elastogenic inducer as TGF-β-bound recombinant LTBP-4. Without LTBP-4, fibulin-5 and tropoelastin deposition was discontinuous and punctate in vitro and in vivo. These data suggest a unique function for LTBP-4 during elastic fibrogenesis, making it a potential therapeutic target for elastic fiber regeneration.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Latent TGF-beta-binding protein 2 binds to DANCE/fibulin-5 and regulates elastic fiber assembly.EMBO J. 2007 Jul 25;26(14):3283-95. doi: 10.1038/sj.emboj.7601768. Epub 2007 Jun 21. EMBO J. 2007. PMID: 17581631 Free PMC article.
-
Fibulin-4 exerts a dual role in LTBP-4L-mediated matrix assembly and function.Proc Natl Acad Sci U S A. 2019 Oct 8;116(41):20428-20437. doi: 10.1073/pnas.1901048116. Epub 2019 Sep 23. Proc Natl Acad Sci U S A. 2019. PMID: 31548410 Free PMC article.
-
LTBP-2 competes with tropoelastin for binding to fibulin-5 and heparin, and is a negative modulator of elastinogenesis.Matrix Biol. 2014 Feb;34:114-23. doi: 10.1016/j.matbio.2013.10.007. Epub 2013 Oct 19. Matrix Biol. 2014. PMID: 24148803
-
Recent updates on the molecular network of elastic fiber formation.Essays Biochem. 2019 Sep 13;63(3):365-376. doi: 10.1042/EBC20180052. Print 2019 Sep 13. Essays Biochem. 2019. PMID: 31395654 Review.
-
[Essential role of LTBP-4 in elastic fiber assembly].Seikagaku. 2014 Dec;86(6):770-3. Seikagaku. 2014. PMID: 25675815 Review. Japanese. No abstract available.
Cited by
-
Mechanisms underlying the inhibitory effects of probucol on elastase-induced abdominal aortic aneurysm in mice.Br J Pharmacol. 2020 Jan;177(1):204-216. doi: 10.1111/bph.14857. Epub 2019 Nov 3. Br J Pharmacol. 2020. PMID: 31478560 Free PMC article.
-
Role of fibrilins in human cancer: A narrative review.Health Sci Rep. 2023 Jul 18;6(7):e1434. doi: 10.1002/hsr2.1434. eCollection 2023 Jul. Health Sci Rep. 2023. PMID: 37469709 Free PMC article.
-
Cutis laxa: intersection of elastic fiber biogenesis, TGFβ signaling, the secretory pathway and metabolism.Matrix Biol. 2014 Jan;33:16-22. doi: 10.1016/j.matbio.2013.07.006. Epub 2013 Aug 16. Matrix Biol. 2014. PMID: 23954411 Free PMC article. Review.
-
Crossing Bridges between Extra- and Intra-Cellular Events in Thoracic Aortic Aneurysms.J Atheroscler Thromb. 2018 Feb 1;25(2):99-110. doi: 10.5551/jat.RV17015. Epub 2017 Sep 22. J Atheroscler Thromb. 2018. PMID: 28943527 Free PMC article. Review.
-
Genomic and physiological analyses of the zebrafish atrioventricular canal reveal molecular building blocks of the secondary pacemaker region.Cell Mol Life Sci. 2021 Oct;78(19-20):6669-6687. doi: 10.1007/s00018-021-03939-y. Epub 2021 Sep 23. Cell Mol Life Sci. 2021. PMID: 34557935 Free PMC article.
References
-
- Rosenbloom J, Abrams WR, Mecham R. Extracellular matrix 4: The elastic fiber. FASEB J. 1993;7(13):1208–1218. - PubMed
-
- Kielty CM, Sherratt MJ, Shuttleworth CA. Elastic fibres. J Cell Sci. 2002;115(Pt 14):2817–2828. - PubMed
-
- Wagenseil JE, Mecham RP. New insights into elastic fiber assembly. Birth Defects Res C Embryo Today. 2007;81(4):229–240. - PubMed
-
- Sato F, et al. Distinct steps of cross-linking, self-association, and maturation of tropoelastin are necessary for elastic fiber formation. J Mol Biol. 2007;369(3):841–851. - PubMed
-
- Ramirez F, Sakai LY, Dietz HC, Rifkin DB. Fibrillin microfibrils: Multipurpose extracellular networks in organismal physiology. Physiol Genomics. 2004;19(2):151–154. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

