Evaluation of the therapeutic potential of a CNP analog in a Fgfr3 mouse model recapitulating achondroplasia
- PMID: 23200862
- PMCID: PMC3516592
- DOI: 10.1016/j.ajhg.2012.10.014
Evaluation of the therapeutic potential of a CNP analog in a Fgfr3 mouse model recapitulating achondroplasia
Abstract
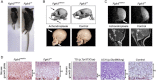
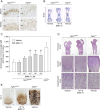
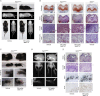
Achondroplasia (ACH), the most common form of dwarfism, is an inherited autosomal-dominant chondrodysplasia caused by a gain-of-function mutation in fibroblast-growth-factor-receptor 3 (FGFR3). C-type natriuretic peptide (CNP) antagonizes FGFR3 downstream signaling by inhibiting the pathway of mitogen-activated protein kinase (MAPK). Here, we report the pharmacological activity of a 39 amino acid CNP analog (BMN 111) with an extended plasma half-life due to its resistance to neutral-endopeptidase (NEP) digestion. In ACH human growth-plate chondrocytes, we demonstrated a decrease in the phosphorylation of extracellular-signal-regulated kinases 1 and 2, confirming that this CNP analog inhibits fibroblast-growth-factor-mediated MAPK activation. Concomitantly, we analyzed the phenotype of Fgfr3(Y367C/+) mice and showed the presence of ACH-related clinical features in this mouse model. We found that in Fgfr3(Y367C/+) mice, treatment with this CNP analog led to a significant recovery of bone growth. We observed an increase in the axial and appendicular skeleton lengths, and improvements in dwarfism-related clinical features included flattening of the skull, reduced crossbite, straightening of the tibias and femurs, and correction of the growth-plate defect. Thus, our results provide the proof of concept that BMN 111, a NEP-resistant CNP analog, might benefit individuals with ACH and hypochondroplasia.
Copyright © 2012 The American Society of Human Genetics. Published by Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Neutral endopeptidase-resistant C-type natriuretic peptide variant represents a new therapeutic approach for treatment of fibroblast growth factor receptor 3-related dwarfism.J Pharmacol Exp Ther. 2015 Apr;353(1):132-49. doi: 10.1124/jpet.114.218560. Epub 2015 Feb 3. J Pharmacol Exp Ther. 2015. PMID: 25650377
-
C-Type Natriuretic Peptide Analog as Therapy for Achondroplasia.Endocr Dev. 2016;30:98-105. doi: 10.1159/000439334. Epub 2015 Dec 10. Endocr Dev. 2016. PMID: 26684019 Review.
-
Phosphatase inhibition by LB-100 enhances BMN-111 stimulation of bone growth.JCI Insight. 2021 May 10;6(9):e141426. doi: 10.1172/jci.insight.141426. JCI Insight. 2021. PMID: 33986191 Free PMC article.
-
Circulatory CNP Rescues Craniofacial Hypoplasia in Achondroplasia.J Dent Res. 2017 Dec;96(13):1526-1534. doi: 10.1177/0022034517716437. Epub 2017 Jun 23. J Dent Res. 2017. PMID: 28644737
-
Vosoritide: First Approval.Drugs. 2021 Nov;81(17):2057-2062. doi: 10.1007/s40265-021-01623-w. Drugs. 2021. PMID: 34694597 Review.
Cited by
-
Rationale, design, and methods of a randomized, controlled, open-label clinical trial with open-label extension to investigate the safety of vosoritide in infants, and young children with achondroplasia at risk of requiring cervicomedullary decompression surgery.Sci Prog. 2021 Jan-Mar;104(1):368504211003782. doi: 10.1177/00368504211003782. Sci Prog. 2021. PMID: 33761804 Free PMC article. Clinical Trial.
-
Current and Emerging Therapeutic Options for the Management of Rare Skeletal Diseases.Paediatr Drugs. 2019 Apr;21(2):95-106. doi: 10.1007/s40272-019-00330-0. Paediatr Drugs. 2019. PMID: 30941653 Review.
-
Novel therapeutic perspectives in Noonan syndrome and RASopathies.Eur J Pediatr. 2024 Mar;183(3):1011-1019. doi: 10.1007/s00431-023-05263-y. Epub 2023 Oct 21. Eur J Pediatr. 2024. PMID: 37863846 Free PMC article. Review.
-
Etiology and Treatment of Growth Delay in Noonan Syndrome.Front Endocrinol (Lausanne). 2021 Jun 4;12:691240. doi: 10.3389/fendo.2021.691240. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 34149626 Free PMC article. Review.
-
Statin treatment rescues FGFR3 skeletal dysplasia phenotypes.Nature. 2014 Sep 25;513(7519):507-11. doi: 10.1038/nature13775. Epub 2014 Sep 17. Nature. 2014. PMID: 25231866
References
-
- Rousseau F., Bonaventure J., Legeai-Mallet L., Pelet A., Rozet J.M., Maroteaux P., Le Merrer M., Munnich A. Mutations in the gene encoding fibroblast growth factor receptor-3 in achondroplasia. Nature. 1994;371:252–254. - PubMed
-
- Shiang R., Thompson L.M., Zhu Y.Z., Church D.M., Fielder T.J., Bocian M., Winokur S.T., Wasmuth J.J. Mutations in the transmembrane domain of FGFR3 cause the most common genetic form of dwarfism, achondroplasia. Cell. 1994;78:335–342. - PubMed
-
- Horton W.A., Hall J.G., Hecht J.T. Achondroplasia. Lancet. 2007;370:162–172. - PubMed
-
- Laederich M.B., Horton W.A. Achondroplasia: Pathogenesis and implications for future treatment. Curr. Opin. Pediatr. 2010;22:516–523. - PubMed
-
- Jonquoy A., Mugniery E., Benoist-Lasselin C., Kaci N., Le Corre L., Barbault F., Girard A.L., Le Merrer Y., Busca P., Schibler L. A novel tyrosine kinase inhibitor restores chondrocyte differentiation and promotes bone growth in a gain-of-function Fgfr3 mouse model. Hum. Mol. Genet. 2012;21:841–851. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

