Multiple novel nesprin-1 and nesprin-2 variants act as versatile tissue-specific intracellular scaffolds
- PMID: 22768332
- PMCID: PMC3388047
- DOI: 10.1371/journal.pone.0040098
Multiple novel nesprin-1 and nesprin-2 variants act as versatile tissue-specific intracellular scaffolds
Abstract
Background: Nesprins (Nuclear envelope spectrin-repeat proteins) are a novel family of giant spectrin-repeat containing proteins. The nesprin-1 and nesprin-2 genes consist of 146 and 116 exons which encode proteins of ∼1mDa and ∼800 kDa is size respectively when all the exons are utilised in translation. However emerging data suggests that the nesprins have multiple alternative start and termination sites throughout their genes allowing the generation of smaller isoforms.
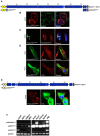
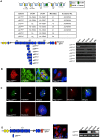
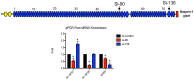
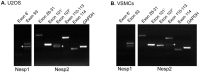
Results: In this study we set out to identify novel alternatively transcribed nesprin variants by screening the EST database and by using RACE analysis to identify cDNA ends. These two methods provided potential hits for alternative start and termination sites that were validated by PCR and DNA sequencing. We show that these alternative sites are not only expressed in a tissue specific manner but by combining different sites together it is possible to create a wide array of nesprin variants. By cloning and expressing small novel nesprin variants into human fibroblasts and U2OS cells we show localization to actin stress-fibres, focal adhesions, microtubules, the nucleolus, nuclear matrix and the nuclear envelope (NE). Furthermore we show that the sub-cellular localization of individual nesprin variants can vary depending on the cell type, suggesting any single nesprin variant may have different functions in different cell types.
Conclusions: These studies suggest nesprins act as highly versatile tissue specific intracellular protein scaffolds and identify potential novel functions for nesprins beyond cytoplasmic-nuclear coupling. These alternate functions may also account for the diverse range of disease phenotypes observed when these genes are mutated.
Conflict of interest statement
Figures



Similar articles
-
Nesprin-2 Giant (NUANCE) maintains nuclear envelope architecture and composition in skin.J Cell Sci. 2008 Jun 1;121(11):1887-98. doi: 10.1242/jcs.019075. Epub 2008 May 13. J Cell Sci. 2008. PMID: 18477613
-
Nesprins: tissue-specific expression of epsilon and other short isoforms.PLoS One. 2014 Apr 9;9(4):e94380. doi: 10.1371/journal.pone.0094380. eCollection 2014. PLoS One. 2014. PMID: 24718612 Free PMC article.
-
Nesprins, but not sun proteins, switch isoforms at the nuclear envelope during muscle development.Dev Dyn. 2010 Mar;239(3):998-1009. doi: 10.1002/dvdy.22229. Dev Dyn. 2010. PMID: 20108321 Free PMC article.
-
Nesprins in health and disease.Semin Cell Dev Biol. 2014 May;29:169-79. doi: 10.1016/j.semcdb.2013.12.010. Epub 2013 Dec 25. Semin Cell Dev Biol. 2014. PMID: 24374011 Review.
-
Nesprins: from the nuclear envelope and beyond.Expert Rev Mol Med. 2013 Jul 5;15:e5. doi: 10.1017/erm.2013.6. Expert Rev Mol Med. 2013. PMID: 23830188 Free PMC article. Review.
Cited by
-
Mutations in Hedgehog pathway genes in fetal rhabdomyomas.J Pathol. 2013 Sep;231(1):44-52. doi: 10.1002/path.4229. J Pathol. 2013. PMID: 23780909 Free PMC article.
-
Mapping the micro-proteome of the nuclear lamina and lamina-associated domains.Life Sci Alliance. 2021 Mar 23;4(5):e202000774. doi: 10.26508/lsa.202000774. Print 2021 May. Life Sci Alliance. 2021. PMID: 33758005 Free PMC article.
-
Apoptosis-induced translocation of nesprin-2 from the nuclear envelope to mitochondria is associated with mitochondrial dysfunction.Nucleus. 2024 Dec;15(1):2413501. doi: 10.1080/19491034.2024.2413501. Epub 2024 Oct 15. Nucleus. 2024. PMID: 39402980 Free PMC article.
-
Cellular mechanosensing: getting to the nucleus of it all.Prog Biophys Mol Biol. 2014 Aug;115(2-3):76-92. doi: 10.1016/j.pbiomolbio.2014.06.009. Epub 2014 Jul 5. Prog Biophys Mol Biol. 2014. PMID: 25008017 Free PMC article. Review.
-
Syne2b/Nesprin-2 Is Required for Actin Organization and Epithelial Integrity During Epiboly Movement in Zebrafish.Front Cell Dev Biol. 2021 Jun 17;9:671887. doi: 10.3389/fcell.2021.671887. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34222245 Free PMC article.
References
-
- Zhang Q, Skepper JN, Yang F, Davies JD, Hegyi L, et al. Nesprins: a novel family of spectrin-repeat-containing proteins that localize to the nuclear membrane in multiple tissues. J Cell Sci. 2001;114:4485–4498. - PubMed
-
- Zhang Q, Ragnauth CD, Skepper JN, Worth NF, Warren DT, et al. Nesprin-2 is a multi-isomeric protein that binds lamin and emerin at the nuclear envelope and forms a subcellular network in skeletal muscle. J Cell Sci. 2005;118:673–687. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

