Mutations in the RNA exosome component gene EXOSC3 cause pontocerebellar hypoplasia and spinal motor neuron degeneration
- PMID: 22544365
- PMCID: PMC3366034
- DOI: 10.1038/ng.2254
Mutations in the RNA exosome component gene EXOSC3 cause pontocerebellar hypoplasia and spinal motor neuron degeneration
Abstract
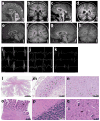
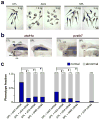
RNA exosomes are multi-subunit complexes conserved throughout evolution and are emerging as the major cellular machinery for processing, surveillance and turnover of a diverse spectrum of coding and noncoding RNA substrates essential for viability. By exome sequencing, we discovered recessive mutations in EXOSC3 (encoding exosome component 3) in four siblings with infantile spinal motor neuron disease, cerebellar atrophy, progressive microcephaly and profound global developmental delay, consistent with pontocerebellar hypoplasia type 1 (PCH1; MIM 607596). We identified mutations in EXOSC3 in an additional 8 of 12 families with PCH1. Morpholino knockdown of exosc3 in zebrafish embryos caused embryonic maldevelopment, resulting in small brain size and poor motility, reminiscent of human clinical features, and these defects were largely rescued by co-injection with wild-type but not mutant exosc3 mRNA. These findings represent the first example of an RNA exosome core component gene that is responsible for a human disease and further implicate dysregulation of RNA processing in cerebellar and spinal motor neuron maldevelopment and degeneration.
Conflict of interest statement
The authors declare no competing financial interests.
Figures

Similar articles
-
Variants in EXOSC9 Disrupt the RNA Exosome and Result in Cerebellar Atrophy with Spinal Motor Neuronopathy.Am J Hum Genet. 2018 May 3;102(5):858-873. doi: 10.1016/j.ajhg.2018.03.011. Am J Hum Genet. 2018. PMID: 29727687 Free PMC article.
-
EXOSC3 mutations in pontocerebellar hypoplasia type 1: novel mutations and genotype-phenotype correlations.Orphanet J Rare Dis. 2014 Feb 13;9:23. doi: 10.1186/1750-1172-9-23. Orphanet J Rare Dis. 2014. PMID: 24524299 Free PMC article.
-
A Chemical Biology Approach to Model Pontocerebellar Hypoplasia Type 1B (PCH1B).ACS Chem Biol. 2018 Oct 19;13(10):3000-3010. doi: 10.1021/acschembio.8b00745. Epub 2018 Sep 6. ACS Chem Biol. 2018. PMID: 30141626 Free PMC article.
-
Pontocerebellar hypoplasia type 1 for the neuropediatrician: Genotype-phenotype correlations and diagnostic guidelines based on new cases and overview of the literature.Eur J Paediatr Neurol. 2018 Jul;22(4):674-681. doi: 10.1016/j.ejpn.2018.03.011. Epub 2018 Apr 3. Eur J Paediatr Neurol. 2018. PMID: 29656927 Review.
-
The RNA exosome and RNA exosome-linked disease.RNA. 2018 Feb;24(2):127-142. doi: 10.1261/rna.064626.117. Epub 2017 Nov 1. RNA. 2018. PMID: 29093021 Free PMC article. Review.
Cited by
-
Midbrain and hindbrain malformations: advances in clinical diagnosis, imaging, and genetics.Lancet Neurol. 2013 Apr;12(4):381-93. doi: 10.1016/S1474-4422(13)70024-3. Epub 2013 Mar 18. Lancet Neurol. 2013. PMID: 23518331 Free PMC article. Review.
-
The genetics of cerebellar malformations.Semin Fetal Neonatal Med. 2016 Oct;21(5):321-32. doi: 10.1016/j.siny.2016.04.008. Epub 2016 May 7. Semin Fetal Neonatal Med. 2016. PMID: 27160001 Free PMC article. Review.
-
Nuclear RNA Exosome and Pervasive Transcription: Dual Sculptors of Genome Function.Int J Mol Sci. 2021 Dec 13;22(24):13401. doi: 10.3390/ijms222413401. Int J Mol Sci. 2021. PMID: 34948199 Free PMC article. Review.
-
Variants in EXOSC9 Disrupt the RNA Exosome and Result in Cerebellar Atrophy with Spinal Motor Neuronopathy.Am J Hum Genet. 2018 May 3;102(5):858-873. doi: 10.1016/j.ajhg.2018.03.011. Am J Hum Genet. 2018. PMID: 29727687 Free PMC article.
-
Competition between maturation and degradation drives human snRNA 3' end quality control.Genes Dev. 2020 Jul 1;34(13-14):989-1001. doi: 10.1101/gad.336891.120. Epub 2020 Jun 4. Genes Dev. 2020. PMID: 32499401 Free PMC article.
References
-
- Mitchell P, Petfalski E, Shevchenko A, Mann M, Tollervey D. The exosome: a conserved eukaryotic RNA processing complex containing multiple 3′-->5′ exoribonucleases. Cell. 1997;91:457–466. - PubMed
-
- Jensen THe. RNA Exosome. Adv Exp Med Biol. 2010;702 - PubMed
-
- de Leon GA, Grover WD, D’Cruz CA. Amyotrophic cerebellar hypoplasia: a specific form of infantile spinal atrophy. Acta Neuropathol. 1984;63:282–286. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

