Mutations in the cardiac L-type calcium channel associated with inherited J-wave syndromes and sudden cardiac death
- PMID: 20817017
- PMCID: PMC2999985
- DOI: 10.1016/j.hrthm.2010.08.026
Mutations in the cardiac L-type calcium channel associated with inherited J-wave syndromes and sudden cardiac death
Abstract
Background: L-type calcium channel (LTCC) mutations have been associated with Brugada syndrome (BrS), short QT (SQT) syndrome, and Timothy syndrome (LQT8). Little is known about the extent to which LTCC mutations contribute to the J-wave syndromes associated with sudden cardiac death.
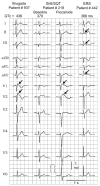
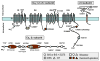
Objective: The purpose of this study was to identify mutations in the α1, β2, and α2δ subunits of LTCC (Ca(v)1.2) among 205 probands diagnosed with BrS, idiopathic ventricular fibrillation (IVF), and early repolarization syndrome (ERS). CACNA1C, CACNB2b, and CACNA2D1 genes of 162 probands with BrS and BrS+SQT, 19 with IVF, and 24 with ERS were screened by direct sequencing.
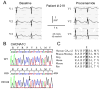
Methods/results: Overall, 23 distinct mutations were identified. A total of 12.3%, 5.2%, and 16% of BrS/BrS+SQT, IVF, and ERS probands displayed mutations in α1, β2, and α2δ subunits of LTCC, respectively. When rare polymorphisms were included, the yield increased to 17.9%, 21%, and 29.1% for BrS/BrS+SQT, IVF, and ERS probands, respectively. Functional expression of two CACNA1C mutations associated with BrS and BrS+SQT led to loss of function in calcium channel current. BrS probands displaying a normal QTc had additional variations known to prolong the QT interval.
Conclusion: The study results indicate that mutations in the LTCCs are detected in a high percentage of probands with J-wave syndromes associated with inherited cardiac arrhythmias, suggesting that genetic screening of Ca(v) genes may be a valuable diagnostic tool in identifying individuals at risk. These results are the first to identify CACNA2D1 as a novel BrS susceptibility gene and CACNA1C, CACNB2, and CACNA2D1 as possible novel ERS susceptibility genes.
Copyright © 2010 Heart Rhythm Society. Published by Elsevier Inc. All rights reserved.
Figures
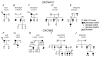

Comment in
-
Exercise extreme caution when calling rare genetic variants novel arrhythmia syndrome susceptibility mutations.Heart Rhythm. 2010 Dec;7(12):1883-5. doi: 10.1016/j.hrthm.2010.10.008. Epub 2010 Oct 13. Heart Rhythm. 2010. PMID: 20950711 No abstract available.
Similar articles
-
Loss-of-function mutations in the cardiac calcium channel underlie a new clinical entity characterized by ST-segment elevation, short QT intervals, and sudden cardiac death.Circulation. 2007 Jan 30;115(4):442-9. doi: 10.1161/CIRCULATIONAHA.106.668392. Epub 2007 Jan 15. Circulation. 2007. PMID: 17224476 Free PMC article.
-
L-type calcium channel mutations in Japanese patients with inherited arrhythmias.Circ J. 2013;77(7):1799-806. doi: 10.1253/circj.cj-12-1457. Epub 2013 Apr 11. Circ J. 2013. PMID: 23575362 Clinical Trial.
-
An African loss-of-function CACNA1C variant p.T1787M associated with a risk of ventricular fibrillation.Sci Rep. 2018 Oct 2;8(1):14619. doi: 10.1038/s41598-018-32867-4. Sci Rep. 2018. PMID: 30279520 Free PMC article.
-
Mutations in voltage-gated L-type calcium channel: implications in cardiac arrhythmia.Channels (Austin). 2018;12(1):201-218. doi: 10.1080/19336950.2018.1499368. Channels (Austin). 2018. PMID: 30027834 Free PMC article. Review.
-
The genetic basis of Brugada syndrome: a mutation update.Hum Mutat. 2009 Sep;30(9):1256-66. doi: 10.1002/humu.21066. Hum Mutat. 2009. PMID: 19606473 Review.
Cited by
-
Molecular genetics and functional anomalies in a series of 248 Brugada cases with 11 mutations in the TRPM4 channel.PLoS One. 2013;8(1):e54131. doi: 10.1371/journal.pone.0054131. Epub 2013 Jan 30. PLoS One. 2013. PMID: 23382873 Free PMC article.
-
Genetic etiology and evaluation of sudden cardiac death.Curr Cardiol Rep. 2013 Aug;15(8):389. doi: 10.1007/s11886-013-0389-8. Curr Cardiol Rep. 2013. PMID: 23812838 Review.
-
Brugada Syndrome and Early Repolarisation: Distinct Clinical Entities or Different Phenotypes of the Same Genetic Disease?Arrhythm Electrophysiol Rev. 2016 Aug;5(2):84-9. doi: 10.15420/AER.2016.23.2. Arrhythm Electrophysiol Rev. 2016. PMID: 27617086 Free PMC article.
-
Distinct Features of Probands With Early Repolarization and Brugada Syndromes Carrying SCN5A Pathogenic Variants.J Am Coll Cardiol. 2021 Oct 19;78(16):1603-1617. doi: 10.1016/j.jacc.2021.08.024. J Am Coll Cardiol. 2021. PMID: 34649698 Free PMC article.
-
Evaluating the Impact of Sex and Gender in Brugada Syndrome.J Innov Card Rhythm Manag. 2019 Feb 15;10(2):3530-3535. doi: 10.19102/icrm.2019.100205. eCollection 2019 Feb. J Innov Card Rhythm Manag. 2019. PMID: 32479580 Free PMC article. No abstract available.
References
-
- Priori SG, Aliot E, Blomstrom-Lundqvist C, et al. Task Force on Sudden Cardiac Death, European Society of Cardiology. Europace. 2002;4:3–18. - PubMed
-
- Behr ER, Dalageorgou C, Christiansen M, et al. Sudden arrhythmic death syndrome: familial evaluation identifies inheritable heart disease in the majority of families. Eur Heart J. 2008;29:1670–1680. - PubMed
-
- Hedley PL, Jorgensen P, Schlamowitz S, et al. The genetic basis of Brugada syndrome: a mutation update. Hum Mutat. 2009;30:1256–1266. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

