Mutations in LTBP4 cause a syndrome of impaired pulmonary, gastrointestinal, genitourinary, musculoskeletal, and dermal development
- PMID: 19836010
- PMCID: PMC2775835
- DOI: 10.1016/j.ajhg.2009.09.013
Mutations in LTBP4 cause a syndrome of impaired pulmonary, gastrointestinal, genitourinary, musculoskeletal, and dermal development
Abstract
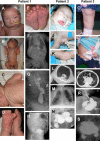
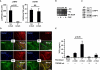
We report recessive mutations in the gene for the latent transforming growth factor-beta binding protein 4 (LTBP4) in four unrelated patients with a human syndrome disrupting pulmonary, gastrointestinal, urinary, musculoskeletal, craniofacial, and dermal development. All patients had severe respiratory distress, with cystic and atelectatic changes in the lungs complicated by tracheomalacia and diaphragmatic hernia. Three of the four patients died of respiratory failure. Cardiovascular lesions were mild, limited to pulmonary artery stenosis and patent foramen ovale. Gastrointestinal malformations included diverticulosis, enlargement, tortuosity, and stenosis at various levels of the intestinal tract. The urinary tract was affected by diverticulosis and hydronephrosis. Joint laxity and low muscle tone contributed to musculoskeletal problems compounded by postnatal growth delay. Craniofacial features included microretrognathia, flat midface, receding forehead, and wide fontanelles. All patients had cutis laxa. Four of the five identified LTBP4 mutations led to premature termination of translation and destabilization of the LTBP4 mRNA. Impaired synthesis and lack of deposition of LTBP4 into the extracellular matrix (ECM) caused increased transforming growth factor-beta (TGF-beta) activity in cultured fibroblasts and defective elastic fiber assembly in all tissues affected by the disease. These molecular defects were associated with blocked alveolarization and airway collapse in the lung. Our results show that coupling of TGF-beta signaling and ECM assembly is essential for proper development and is achieved in multiple human organ systems by multifunctional proteins such as LTBP4.
Figures


Similar articles
-
Comprehensive clinical and molecular analysis of 12 families with type 1 recessive cutis laxa.Hum Mutat. 2013 Jan;34(1):111-21. doi: 10.1002/humu.22165. Epub 2012 Aug 13. Hum Mutat. 2013. PMID: 22829427 Free PMC article.
-
Clinical and molecular characterization of an 18-month-old infant with autosomal recessive cutis laxa type 1C due to a novel LTBP4 pathogenic variant, and literature review.Mol Genet Genomic Med. 2019 Jul;7(7):e00735. doi: 10.1002/mgg3.735. Epub 2019 May 21. Mol Genet Genomic Med. 2019. PMID: 31115174 Free PMC article. Review.
-
Bi-allelic premature truncating variants in LTBP1 cause cutis laxa syndrome.Am J Hum Genet. 2021 Jun 3;108(6):1095-1114. doi: 10.1016/j.ajhg.2021.04.016. Epub 2021 May 14. Am J Hum Genet. 2021. PMID: 33991472 Free PMC article.
-
Latent transforming growth factor binding protein 4 regulates transforming growth factor beta receptor stability.Hum Mol Genet. 2015 Jul 15;24(14):4024-36. doi: 10.1093/hmg/ddv139. Epub 2015 Apr 16. Hum Mol Genet. 2015. PMID: 25882708 Free PMC article.
-
Autosomal recessive cutis laxa type 1C with a homozygous LTBP4 splicing variant: a case report and update of literature.Mol Biol Rep. 2022 May;49(5):4135-4140. doi: 10.1007/s11033-022-07454-6. Epub 2022 Apr 20. Mol Biol Rep. 2022. PMID: 35445908 Review.
Cited by
-
The complexity of elastic fibre biogenesis in the skin--a perspective to the clinical heterogeneity of cutis laxa.Exp Dermatol. 2013 Feb;22(2):88-92. doi: 10.1111/exd.12025. Epub 2012 Oct 23. Exp Dermatol. 2013. PMID: 23088642 Free PMC article.
-
Congenic mice provide in vivo evidence for a genetic locus that modulates intrinsic transforming growth factor β1-mediated signaling and bone acquisition.J Bone Miner Res. 2012 Jun;27(6):1345-56. doi: 10.1002/jbmr.1590. J Bone Miner Res. 2012. PMID: 22407846 Free PMC article.
-
LTBPs, more than just an escort service.J Cell Biochem. 2012 Feb;113(2):410-8. doi: 10.1002/jcb.23385. J Cell Biochem. 2012. PMID: 22223425 Free PMC article. Review.
-
Further characterization of ATP6V0A2-related autosomal recessive cutis laxa.Hum Genet. 2012 Nov;131(11):1761-73. doi: 10.1007/s00439-012-1197-8. Epub 2012 Jul 8. Hum Genet. 2012. PMID: 22773132
-
Latent TGF-β binding protein 4 promotes elastic fiber assembly by interacting with fibulin-5.Proc Natl Acad Sci U S A. 2013 Feb 19;110(8):2852-7. doi: 10.1073/pnas.1215779110. Epub 2013 Feb 4. Proc Natl Acad Sci U S A. 2013. PMID: 23382201 Free PMC article.
References
-
- Wu M.Y., Hill C.S. Tgf-beta superfamily signaling in embryonic development and homeostasis. Dev. Cell. 2009;16:329–343. - PubMed
-
- Rifkin D.B. Latent transforming growth factor-beta (TGF-beta) binding proteins: orchestrators of TGF-beta availability. J. Biol. Chem. 2005;280:7409–7412. - PubMed
-
- Annes J.P., Munger J.S., Rifkin D.B. Making sense of latent TGFbeta activation. J. Cell Sci. 2003;116:217–224. - PubMed
-
- Rodriguez-Revenga L., Iranzo P., Badenas C., Puig S., Carrio A., Mila M. A novel elastin gene mutation resulting in an autosomal dominant form of cutis laxa. Arch. Dermatol. 2004;140:1135–1139. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

