Role of the polycomb protein EED in the propagation of repressive histone marks
- PMID: 19767730
- PMCID: PMC3772642
- DOI: 10.1038/nature08398
Role of the polycomb protein EED in the propagation of repressive histone marks
Abstract
Polycomb group proteins have an essential role in the epigenetic maintenance of repressive chromatin states. The gene-silencing activity of the Polycomb repressive complex 2 (PRC2) depends on its ability to trimethylate lysine 27 of histone H3 (H3K27) by the catalytic SET domain of the EZH2 subunit, and at least two other subunits of the complex: SUZ12 and EED. Here we show that the carboxy-terminal domain of EED specifically binds to histone tails carrying trimethyl-lysine residues associated with repressive chromatin marks, and that this leads to the allosteric activation of the methyltransferase activity of PRC2. Mutations in EED that prevent it from recognizing repressive trimethyl-lysine marks abolish the activation of PRC2 in vitro and, in Drosophila, reduce global methylation and disrupt development. These findings suggest a model for the propagation of the H3K27me3 mark that accounts for the maintenance of repressive chromatin domains and for the transmission of a histone modification from mother to daughter cells.
Figures
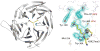
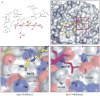

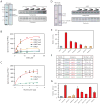

Similar articles
-
Histone methylation by PRC2 is inhibited by active chromatin marks.Mol Cell. 2011 May 6;42(3):330-41. doi: 10.1016/j.molcel.2011.03.025. Mol Cell. 2011. PMID: 21549310
-
Dense chromatin activates Polycomb repressive complex 2 to regulate H3 lysine 27 methylation.Science. 2012 Aug 24;337(6097):971-5. doi: 10.1126/science.1225237. Science. 2012. PMID: 22923582
-
The N terminus of Drosophila ESC binds directly to histone H3 and is required for E(Z)-dependent trimethylation of H3 lysine 27.Mol Cell Biol. 2007 Mar;27(6):2014-26. doi: 10.1128/MCB.01822-06. Epub 2007 Jan 8. Mol Cell Biol. 2007. PMID: 17210640 Free PMC article.
-
Inner workings and regulatory inputs that control Polycomb repressive complex 2.Chromosoma. 2012 Jun;121(3):221-34. doi: 10.1007/s00412-012-0361-1. Epub 2012 Feb 19. Chromosoma. 2012. PMID: 22349693 Free PMC article. Review.
-
The functions of E(Z)/EZH2-mediated methylation of lysine 27 in histone H3.Curr Opin Genet Dev. 2004 Apr;14(2):155-64. doi: 10.1016/j.gde.2004.02.001. Curr Opin Genet Dev. 2004. PMID: 15196462 Review.
Cited by
-
Histone methylation: a dynamic mark in health, disease and inheritance.Nat Rev Genet. 2012 Apr 3;13(5):343-57. doi: 10.1038/nrg3173. Nat Rev Genet. 2012. PMID: 22473383 Free PMC article. Review.
-
Forecasting histone methylation by Polycomb complexes with minute-scale precision.Sci Adv. 2023 Dec 22;9(51):eadj8198. doi: 10.1126/sciadv.adj8198. Epub 2023 Dec 22. Sci Adv. 2023. PMID: 38134278 Free PMC article.
-
EED-mediated histone methylation is critical for CNS myelination and remyelination by inhibiting WNT, BMP, and senescence pathways.Sci Adv. 2020 Aug 12;6(33):eaaz6477. doi: 10.1126/sciadv.aaz6477. eCollection 2020 Aug. Sci Adv. 2020. PMID: 32851157 Free PMC article.
-
Molecular basis of PRC1 targeting to Polycomb response elements by PhoRC.Genes Dev. 2016 May 1;30(9):1116-27. doi: 10.1101/gad.279141.116. Genes Dev. 2016. PMID: 27151979 Free PMC article.
-
Distinctive aspects of the placental epigenome and theories as to how they arise.Cell Mol Life Sci. 2022 Oct 26;79(11):569. doi: 10.1007/s00018-022-04568-9. Cell Mol Life Sci. 2022. PMID: 36287261 Free PMC article. Review.
References
-
- Schuettengruber B, Chourrout D, Vervoort M, Leblanc B, Cavalli G. Genome Regulation by Polycomb and Trithorax Proteins. Cell. 2007;128:735–745. - PubMed
-
- Czermin B, et al. Drosophila enhancer of Zeste/ESC complexes have a histone H3 methyltransferase activity that marks chromosomal Polycomb sites. Cell. 2002;111:185–96. - PubMed
-
- Cao R, et al. Role of histone H3 lysine 27 methylation in Polycomb-group silencing. Science. 2002;298:1039–43. - PubMed
-
- Muller J, et al. Histone methyltransferase activity of a Drosophila Polycomb group repressor complex. Cell. 2002;111:197–208. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

