Inactivation of Escherichia coli endotoxin by soft hydrothermal processing
- PMID: 19502435
- PMCID: PMC2725499
- DOI: 10.1128/AEM.00122-09
Inactivation of Escherichia coli endotoxin by soft hydrothermal processing
Abstract
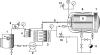
Bacterial endotoxins, also known as lipopolysaccharides, are a fever-producing by-product of gram-negative bacteria commonly known as pyrogens. It is essential to remove endotoxins from parenteral preparations since they have multiple injurious biological activities. Because of their strong heat resistance (e.g., requiring dry-heat sterilization at 250 degrees C for 30 min) and the formation of various supramolecular aggregates, depyrogenation is more difficult than sterilization. We report here that soft hydrothermal processing, which has many advantages in safety and cost efficiency, is sufficient to assure complete depyrogenation by the inactivation of endotoxins. The endotoxin concentration in a sample was measured by using a chromogenic limulus method with an endotoxin-specific limulus reagent. The endotoxin concentration was calculated from a standard curve obtained using a serial dilution of a standard solution. We show that endotoxins were completely inactivated by soft hydrothermal processing at 130 degrees C for 60 min or at 140 degrees C for 30 min in the presence of a high steam saturation ratio or with a flow system. Moreover, it is easy to remove endotoxins from water by soft hydrothermal processing similarly at 130 degrees C for 60 min or at 140 degrees C for 30 min, without any requirement for ultrafiltration, nonselective adsorption with a hydrophobic adsorbent, or an anion exchanger. These findings indicate that soft hydrothermal processing, applied in the presence of a high steam saturation ratio or with a flow system, can inactivate endotoxins and may be useful for the depyrogenation of parenterals, including end products and medical devices that cannot be exposed to the high temperatures of dry heat treatments.
Figures

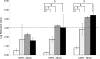
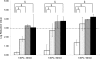
Similar articles
-
Destruction of typical endotoxins by dry heat as determined using LAL assay and pyrogen assay.J Parenter Sci Technol. 1993 Sep-Oct;47(5):258-64. J Parenter Sci Technol. 1993. PMID: 8263663
-
A Validation Study of the Limulus Amebocyte Lysate Test as an End-Product Endotoxin Test for Polyvalent Horse Snake Antivenom.PDA J Pharm Sci Technol. 2019 Nov-Dec;73(6):562-571. doi: 10.5731/pdajpst.2018.009522. Epub 2019 Oct 15. PDA J Pharm Sci Technol. 2019. PMID: 30770487
-
Effect of steam-heat treatment with/without divalent cations on the inactivation of lipopolysaccharides from several bacterial species.PDA J Pharm Sci Technol. 1996 Mar-Apr;50(2):129-35. PDA J Pharm Sci Technol. 1996. PMID: 8935780
-
[Application of a bacterial endotoxin test for parenteral drugs].Eisei Shikenjo Hokoku. 1994;(112):209-11. Eisei Shikenjo Hokoku. 1994. PMID: 8854936 Review. Japanese.
-
Measurement of endotoxin levels in hemoglobin preparations.Methods Enzymol. 1994;231:75-91. doi: 10.1016/0076-6879(94)31008-4. Methods Enzymol. 1994. PMID: 8041288 Review. No abstract available.
Cited by
-
Effect of Food Endotoxin on Infant Health.Toxins (Basel). 2021 Apr 22;13(5):298. doi: 10.3390/toxins13050298. Toxins (Basel). 2021. PMID: 33922125 Free PMC article. Review.
-
Cloning, Secretory Expression and Characterization of a Unique pH-Stable and Cold-Adapted Alginate Lyase.Mar Drugs. 2020 Apr 1;18(4):189. doi: 10.3390/md18040189. Mar Drugs. 2020. PMID: 32244721 Free PMC article.
-
Low endotoxin E. coli strain-derived plasmids reduce rAAV vector-mediated immune responses both in vitro and in vivo.Mol Ther Methods Clin Dev. 2021 Jun 24;22:293-303. doi: 10.1016/j.omtm.2021.06.009. eCollection 2021 Sep 10. Mol Ther Methods Clin Dev. 2021. PMID: 34485612 Free PMC article.
-
Pre-clinical immunotoxicity studies of nanotechnology-formulated drugs: Challenges, considerations and strategy.J Control Release. 2015 Dec 28;220(Pt B):571-83. doi: 10.1016/j.jconrel.2015.08.056. Epub 2015 Sep 5. J Control Release. 2015. PMID: 26348388 Free PMC article. Review.
-
Advanced Proteomics as a Powerful Tool for Studying Toxins of Human Bacterial Pathogens.Toxins (Basel). 2019 Oct 4;11(10):576. doi: 10.3390/toxins11100576. Toxins (Basel). 2019. PMID: 31590258 Free PMC article. Review.
References
-
- Andersson, T., K. Hartonen, T. Hyotylainen, and M. L. Riekkola. 2002. Pressurized hot water extraction and thermal desorption of polycyclic aromatic hydrocarbons from sediment with use of a novel extraction vessel. Anal. Chim. Acta 466:93-100.
-
- Anspach, F. B. 2001. Endotoxin removal by affinity sorbents. J. Biochem. Biophys. Methods 49:665-681. - PubMed
-
- Bamba, T., R. Matsui, and K. Watabe. 1996. Effect of steam-heat treatment with/without divalent cations on the inactivation of lipopolysaccharides from several bacterial species. PDA J. Pharm. Sci. Technol. 50:129-135. - PubMed
-
- Bamba, T., R. Matsui, and K. Watabe. 1996. Enhancing effect of non-ionic surfactant on the inactivation of lipopolysaccharides by steam-heat treatment. PDA J. Pharm. Sci. Technol. 50:360-365. - PubMed
-
- Bamba, T., R. Matsui, and K. Watabe, and M. Tadanori. 1997. Enhancing effect of non-ionic surfactant on the inactivation of lipopolysaccharides by steam-heat treatment. PDA J. Pharm. Sci. Technol. 51:156-160. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

