Mg2+ imparts NMDA receptor subtype selectivity to the Alzheimer's drug memantine
- PMID: 19261873
- PMCID: PMC2679254
- DOI: 10.1523/JNEUROSCI.3703-08.2009
Mg2+ imparts NMDA receptor subtype selectivity to the Alzheimer's drug memantine
Abstract
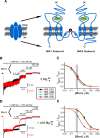
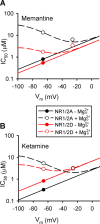
N-methyl-D-aspartate receptors (NMDARs) mediate interneuronal communication and are broadly involved in nervous system physiology and pathology (Dingledine et al., 1999). Memantine, a drug that blocks the ion channel formed by NMDARs, is a widely prescribed treatment of Alzheimer's disease (Schmitt, 2005; Lipton, 2006; Parsons et al., 2007). Research on memantine's mechanism of action has focused on the NMDAR subtypes most highly expressed in adult cerebral cortex, NR1/2A and NR1/2B receptors (Cull-Candy and Leszkiewicz, 2004), and has largely ignored interactions with extracellular Mg(2+) (Mg(2+)(o)). Mg(2+)(o) is an endogenous NMDAR channel blocker that binds near memantine's binding site (Kashiwagi et al., 2002; Chen and Lipton, 2005). We report that a physiological concentration (1 mM) of Mg(2+)(o) decreased memantine inhibition of NR1/2A and NR1/2B receptors nearly 20-fold at a membrane voltage near rest. In contrast, memantine inhibition of the other principal NMDAR subtypes, NR1/2C and NR1/2D receptors, was decreased only approximately 3-fold. As a result, therapeutic memantine concentrations should have negligible effects on NR1/2A or NR1/2B receptor activity but pronounced effects on NR1/2C and NR1/2D receptors. Quantitative modeling showed that the voltage dependence of memantine inhibition also is altered by 1 mM Mg(2+)(o). We report similar results with the NMDAR channel blocker ketamine, a drug used to model schizophrenia (Krystal et al., 2003). These results suggest that currently hypothesized mechanisms of memantine and ketamine action should be reconsidered and that NR1/2C and/or NR1/2D receptors play a more important role in cortical physiology and pathology than previously appreciated.
Figures

Similar articles
-
Memantine binding to a superficial site on NMDA receptors contributes to partial trapping.J Physiol. 2009 Oct 1;587(Pt 19):4589-604. doi: 10.1113/jphysiol.2009.176297. Epub 2009 Aug 17. J Physiol. 2009. PMID: 19687120 Free PMC article.
-
Memantine and Ketamine Differentially Alter NMDA Receptor Desensitization.J Neurosci. 2017 Oct 4;37(40):9686-9704. doi: 10.1523/JNEUROSCI.1173-17.2017. Epub 2017 Sep 6. J Neurosci. 2017. PMID: 28877967 Free PMC article.
-
Effects of Mg2+ on recovery of NMDA receptors from inhibition by memantine and ketamine reveal properties of a second site.Neuropharmacology. 2018 Jul 15;137:344-358. doi: 10.1016/j.neuropharm.2018.05.017. Epub 2018 May 12. Neuropharmacology. 2018. PMID: 29793153 Free PMC article.
-
Mechanism of action of memantine.Curr Opin Pharmacol. 2006 Feb;6(1):61-7. doi: 10.1016/j.coph.2005.09.007. Epub 2005 Dec 20. Curr Opin Pharmacol. 2006. PMID: 16368266 Review.
-
The molecular basis of memantine action in Alzheimer's disease and other neurologic disorders: low-affinity, uncompetitive antagonism.Curr Alzheimer Res. 2005 Apr;2(2):155-65. doi: 10.2174/1567205053585846. Curr Alzheimer Res. 2005. PMID: 15974913 Review.
Cited by
-
Mechanisms of NMDA Receptor Inhibition by Biguanide Compounds.Pharmaceuticals (Basel). 2024 Sep 19;17(9):1234. doi: 10.3390/ph17091234. Pharmaceuticals (Basel). 2024. PMID: 39338396 Free PMC article.
-
GluN2B subunit-containing NMDA receptor antagonists prevent Abeta-mediated synaptic plasticity disruption in vivo.Proc Natl Acad Sci U S A. 2009 Dec 1;106(48):20504-9. doi: 10.1073/pnas.0908083106. Epub 2009 Nov 16. Proc Natl Acad Sci U S A. 2009. PMID: 19918059 Free PMC article.
-
Synaptic versus extrasynaptic NMDA receptor signalling: implications for neurodegenerative disorders.Nat Rev Neurosci. 2010 Oct;11(10):682-96. doi: 10.1038/nrn2911. Epub 2010 Sep 15. Nat Rev Neurosci. 2010. PMID: 20842175 Free PMC article. Review.
-
Potential Roles for the GluN2D NMDA Receptor Subunit in Schizophrenia.Int J Mol Sci. 2023 Jul 23;24(14):11835. doi: 10.3390/ijms241411835. Int J Mol Sci. 2023. PMID: 37511595 Free PMC article. Review.
-
Narrative Review: Low-Dose Ketamine for Pain Management.J Clin Med. 2023 May 2;12(9):3256. doi: 10.3390/jcm12093256. J Clin Med. 2023. PMID: 37176696 Free PMC article. Review.
References
-
- Annetta MG, Iemma D, Garisto C, Tafani C, Proietti R. Ketamine: new indications for an old drug. Curr Drug Targets. 2005;6:789–794. - PubMed
-
- Aracava Y, Pereira EF, Maelicke A, Albuquerque EX. Memantine blocks alpha7* nicotinic acetylcholine receptors more potently than N-methyl-D-aspartate receptors in rat hippocampal neurons. J Pharmacol Exp Ther. 2005;312:1195–1205. - PubMed
-
- Bell KF, Claudio Cuello A. Altered synaptic function in Alzheimer's disease. Eur J Pharmacol. 2006;545:11–21. - PubMed
-
- Blanpied TA, Boeckman FA, Aizenman E, Johnson JW. Trapping channel block of NMDA-activated responses by amantadine and memantine. J Neurophysiol. 1997;77:309–323. - PubMed
-
- Chen HS, Lipton SA. Pharmacological implications of two distinct mechanisms of interaction of memantine with N-methyl-D-aspartate-gated channels. J Pharmacol Exp Ther. 2005;314:961–971. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
