Fatty acid 2-hydroxylase regulates cAMP-induced cell cycle exit in D6P2T schwannoma cells
- PMID: 19171550
- PMCID: PMC2681402
- DOI: 10.1194/jlr.M800666-JLR200
Fatty acid 2-hydroxylase regulates cAMP-induced cell cycle exit in D6P2T schwannoma cells
Abstract
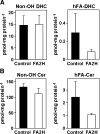
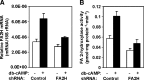
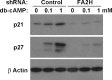
Sphingolipids are ubiquitous components of eukaryotic cells that regulate various cellular functions. In many cell types, a fraction of sphingolipids contain 2-hydroxy fatty acids, produced by fatty acid 2-hydroxylase (FA2H), as the N-acyl chain of ceramide [hydroxyl fatty acid (hFA)-sphingolipids]. FA2H is highly expressed in myelin-forming cells of the nervous system and in epidermal keratinocytes. While hFA-sphingolipids are thought to enhance the physical stability of specialized membranes produced by these cells, physiological significance of hFA-sphingolipids in many other cell types is unknown. In this study, we report novel roles for FA2H and hFA-sphingolipids in the regulation of the cell cycle. Treatment of D6P2T Schwannoma cells with dibutyryl-cAMP (db-cAMP) induced exit from the cell cycle with concomitant upregulation of FA2H. Partial silencing of FA2H in D6P2T cells resulted in 60-70% reduction of hFA-dihydroceramide and hFA-ceramide, with no effect on nonhydroxy dihydroceramide and ceramide. Under these conditions, db-cAMP no longer induced cell cycle exit, and cells continued to grow and divide. Immunoblot analyses revealed that FA2H silencing prevented db-cAMP-induced upregulation of cyclin-dependent kinase inhibitors p21 and p27. These results provide evidence that FA2H is a negative regulator of the cell cycle and facilitates db-cAMP-induced cell cycle exit in D6P2T cells.
Figures

Similar articles
-
Fatty acid 2-Hydroxylation in mammalian sphingolipid biology.Biochim Biophys Acta. 2010 Apr;1801(4):405-14. doi: 10.1016/j.bbalip.2009.12.004. Epub 2009 Dec 21. Biochim Biophys Acta. 2010. PMID: 20026285 Free PMC article. Review.
-
Fatty acid 2-hydroxylase, encoded by FA2H, accounts for differentiation-associated increase in 2-OH ceramides during keratinocyte differentiation.J Biol Chem. 2007 May 4;282(18):13211-9. doi: 10.1074/jbc.M611562200. Epub 2007 Mar 12. J Biol Chem. 2007. PMID: 17355976 Clinical Trial.
-
FA2H is responsible for the formation of 2-hydroxy galactolipids in peripheral nervous system myelin.J Lipid Res. 2008 Jan;49(1):153-61. doi: 10.1194/jlr.M700400-JLR200. Epub 2007 Sep 27. J Lipid Res. 2008. PMID: 17901466 Free PMC article.
-
2'-Hydroxy ceramide in membrane homeostasis and cell signaling.Adv Biol Regul. 2014 Jan;54:223-30. doi: 10.1016/j.jbior.2013.09.012. Epub 2013 Oct 8. Adv Biol Regul. 2014. PMID: 24139861 Free PMC article. Review.
-
FA2H-dependent fatty acid 2-hydroxylation in postnatal mouse brain.J Lipid Res. 2006 Dec;47(12):2772-80. doi: 10.1194/jlr.M600362-JLR200. Epub 2006 Sep 23. J Lipid Res. 2006. PMID: 16998236
Cited by
-
MicroRNA-1297 downregulation inhibits breast cancer cell epithelial-mesenchymal transition and proliferation in a FA2H-dependent manner.Oncol Lett. 2020 Dec;20(6):277. doi: 10.3892/ol.2020.12140. Epub 2020 Sep 23. Oncol Lett. 2020. PMID: 33014155 Free PMC article.
-
Gene expression in triple-negative breast cancer in relation to survival.Breast Cancer Res Treat. 2018 Aug;171(1):199-207. doi: 10.1007/s10549-018-4816-9. Epub 2018 May 10. Breast Cancer Res Treat. 2018. PMID: 29748761 Free PMC article.
-
Neurodegeneration with brain iron accumulation: diagnosis and management.J Mov Disord. 2015 Jan;8(1):1-13. doi: 10.14802/jmd.14034. Epub 2015 Jan 13. J Mov Disord. 2015. PMID: 25614780 Free PMC article. Review.
-
Defective FA2H leads to a novel form of neurodegeneration with brain iron accumulation (NBIA).Ann Neurol. 2010 Nov;68(5):611-8. doi: 10.1002/ana.22122. Ann Neurol. 2010. PMID: 20853438 Free PMC article.
-
Overexpression of HexCer and LacCer containing 2-hydroxylated fatty acids in cholangiocarcinoma and the association of the increase of LacCer (d18:1-h23:0) with shorter survival of the patients.Glycoconj J. 2019 Apr;36(2):103-111. doi: 10.1007/s10719-019-09864-4. Epub 2019 Mar 19. Glycoconj J. 2019. PMID: 30888588
References
-
- Hakomori S. 2003. Structure, organization, and function of glycosphingolipids in membrane. Curr. Opin. Hematol. 10 16–24. - PubMed
-
- Sillence D. J., and F. M. Platt. 2004. Glycosphingolipids in endocytic membrane transport. Semin. Cell Dev. Biol. 15 409–416. - PubMed
-
- Norton, W. T., and W. Cammer. 1984. Isolation and characterization of myelin. In Myelin. P. Morell, editor. Plenum, New York, NY. 147–195.
-
- Downing D. T. 1992. Lipid and protein structures in the permeability barrier of mammalian epidermis. J. Lipid Res. 33 301–313. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

