Targeting of the 5-HT1A serotonin receptor to neuronal dendrites is mediated by Yif1B
- PMID: 18685031
- PMCID: PMC6670764
- DOI: 10.1523/JNEUROSCI.4487-07.2008
Targeting of the 5-HT1A serotonin receptor to neuronal dendrites is mediated by Yif1B
Abstract
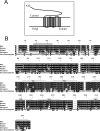
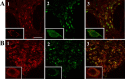
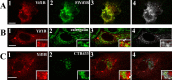
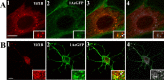
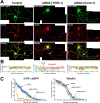
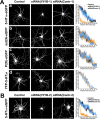
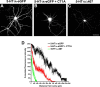
The 5-HT(1A) receptor (5-HT(1A)R) is the most extensively characterized serotonin (5-HT) receptor mainly because of its involvement in the mode of action of antidepressants. The 5-HT(1A)R is confined to the somatodendritic domain of central neurons, where it mediates serotonin-evoked hyperpolarization. Our previous studies underlined the role of the short 5-HT(1A)R C-terminal domain in receptor targeting to dendrites. We used this 17 aa region as bait in a yeast two-hybrid screen, and identified, for the first time, an intracellular protein interacting with the 5-HT(1A)R. This protein is homologous to the yeast Yif1p, previously implicated in vesicular trafficking between the endoplasmic reticulum (ER) and the Golgi apparatus, but not yet characterized in mammals. We confirmed 5-HT(1A)R-Yif1B interaction by glutathione S-transferase pull-down experiments using rat brain extracts and transfected cell lines. Yif1B is highly expressed in the brain, and specifically in raphe 5-HT(1A)R-expressing neurons. Colocalization of Yif1B and 5-HT(1A)R was observed in small vesicles involved in transient intracellular trafficking. Last, inhibition of endogenous expression of Yif1B in primary neuron cultures by small interfering RNA specifically prevented the addressing of 5-HT(1A)R to distal portions of the dendrites, without affecting other receptors, such as sst2A, P2X(2), and 5-HT(3A) receptors. Together, our results provide strong evidence that Yif1B is a member of the ER/Golgi trafficking machinery, which plays a key role in specific targeting of 5-HT(1A)R to the neuronal dendrites. This finding opens up new pathways for the study of 5-HT(1A)R regulation by partner proteins and for the development of novel antidepressant drugs.
Figures


Similar articles
-
A new vesicular scaffolding complex mediates the G-protein-coupled 5-HT1A receptor targeting to neuronal dendrites.J Neurosci. 2012 Oct 10;32(41):14227-41. doi: 10.1523/JNEUROSCI.6329-11.2012. J Neurosci. 2012. PMID: 23055492 Free PMC article.
-
Key role of the 5-HT1A receptor addressing protein Yif1B in serotonin neurotransmission and SSRI treatment.J Psychiatry Neurosci. 2020 Sep 1;45(5):344-355. doi: 10.1503/jpn.190134. J Psychiatry Neurosci. 2020. PMID: 32459080 Free PMC article.
-
Dominant role of the cytosolic C-terminal domain of the rat 5-HT1B receptor in axonal-apical targeting.J Neurosci. 2000 Dec 15;20(24):9111-8. doi: 10.1523/JNEUROSCI.20-24-09111.2000. J Neurosci. 2000. PMID: 11124988 Free PMC article.
-
5-HT1A and 5-HT1B receptor agonists and aggression: a pharmacological challenge of the serotonin deficiency hypothesis.Eur J Pharmacol. 2005 Dec 5;526(1-3):125-39. doi: 10.1016/j.ejphar.2005.09.065. Epub 2005 Nov 28. Eur J Pharmacol. 2005. PMID: 16310183 Review.
-
Serotonin and psychostimulant addiction: focus on 5-HT1A-receptors.Prog Neurobiol. 2007 Feb;81(3):133-78. doi: 10.1016/j.pneurobio.2007.01.001. Epub 2007 Jan 17. Prog Neurobiol. 2007. PMID: 17316955 Review.
Cited by
-
A practical guide to evaluating colocalization in biological microscopy.Am J Physiol Cell Physiol. 2011 Apr;300(4):C723-42. doi: 10.1152/ajpcell.00462.2010. Epub 2011 Jan 5. Am J Physiol Cell Physiol. 2011. PMID: 21209361 Free PMC article.
-
The ufmylation cascade controls COPII recruitment, anterograde transport, and sorting of nascent GPCRs at ER.Sci Adv. 2024 Jun 21;10(25):eadm9216. doi: 10.1126/sciadv.adm9216. Epub 2024 Jun 21. Sci Adv. 2024. PMID: 38905340 Free PMC article.
-
Prognostic and immune regulating roles of YIF1B in Pan-Cancer: a potential target for both survival and therapy response evaluation.Biosci Rep. 2020 Jul 31;40(7):BSR20201384. doi: 10.1042/BSR20201384. Biosci Rep. 2020. PMID: 32648580 Free PMC article.
-
Characteristics and Functions of the Yip1 Domain Family (YIPF), Multi-Span Transmembrane Proteins Mainly Localized to the Golgi Apparatus.Front Cell Dev Biol. 2019 Jul 30;7:130. doi: 10.3389/fcell.2019.00130. eCollection 2019. Front Cell Dev Biol. 2019. PMID: 31417902 Free PMC article. Review.
-
A New Mechanism of Receptor Targeting by Interaction between Two Classes of Ligand-Gated Ion Channels.J Neurosci. 2016 Feb 3;36(5):1456-70. doi: 10.1523/JNEUROSCI.2390-15.2016. J Neurosci. 2016. PMID: 26843630 Free PMC article.
References
-
- Albert PR, Zhou QY, Van Tol HH, Bunzow JR, Civelli O. Cloning, functional expression, and mRNA tissue distribution of the rat 5-hydroxytryptamine1A receptor gene. J Biol Chem. 1990;265:5825–5832. - PubMed
-
- Ango F, Robbe D, Tu JC, Xiao B, Worley PF, Pin JP, Bockaert J, Fagni L. Homer-dependent cell surface expression of metabotropic glutamate receptor type 5 in neurons. Mol Cell Neurosci. 2002;20:323–329. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
