Leukoencephalopathy upon disruption of the chloride channel ClC-2
- PMID: 17567819
- PMCID: PMC6672451
- DOI: 10.1523/JNEUROSCI.0338-07.2007
Leukoencephalopathy upon disruption of the chloride channel ClC-2
Abstract
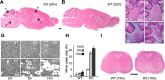
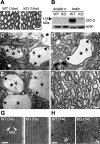
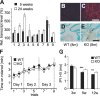
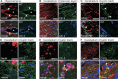
ClC-2 is a broadly expressed plasma membrane chloride channel that is modulated by voltage, cell swelling, and pH. A human mutation leading to a heterozygous loss of ClC-2 has previously been reported to be associated with epilepsy, whereas the disruption of Clcn2 in mice led to testicular and retinal degeneration. We now show that the white matter of the brain and spinal cord of ClC-2 knock-out mice developed widespread vacuolation that progressed with age. Fluid-filled spaces appeared between myelin sheaths of the central but not the peripheral nervous system. Neuronal morphology, in contrast, seemed normal. Except for the previously reported blindness, neurological deficits were mild and included a decreased conduction velocity in neurons of the central auditory pathway. The heterozygous loss of ClC-2 had no detectable functional or morphological consequences. Neither heterozygous nor homozygous ClC-2 knock-out mice had lowered seizure thresholds. Sequencing of a large collection of human DNA and electrophysiological analysis showed that several ClC-2 sequence abnormalities previously found in patients with epilepsy most likely represent innocuous polymorphisms.
Figures
Similar articles
-
Brain white matter oedema due to ClC-2 chloride channel deficiency: an observational analytical study.Lancet Neurol. 2013 Jul;12(7):659-68. doi: 10.1016/S1474-4422(13)70053-X. Epub 2013 May 22. Lancet Neurol. 2013. PMID: 23707145
-
Cellular basis of ClC-2 Cl- channel-related brain and testis pathologies.J Biol Chem. 2021 Jan-Jun;296:100074. doi: 10.1074/jbc.RA120.016031. Epub 2020 Nov 23. J Biol Chem. 2021. PMID: 33187987 Free PMC article.
-
Progressive white matter pathology in the spinal cord of transgenic mice expressing mutant (P301L) human tau.J Neurocytol. 2005 Dec;34(6):397-410. doi: 10.1007/s11068-006-8726-0. Epub 2006 Aug 10. J Neurocytol. 2005. PMID: 16902761
-
[Function of the CLC chloride channels and their implication in human pathology].Nephrologie. 2002;23(3):113-8. Nephrologie. 2002. PMID: 12087807 Review. French.
-
Comparison of CNS and PNS myelin proteins in the pathology of myelin disorders.J Neurol Sci. 2005 Feb 15;228(2):187-9. doi: 10.1016/j.jns.2004.10.005. Epub 2004 Dec 1. J Neurol Sci. 2005. PMID: 15694201 Review. No abstract available.
Cited by
-
De novo and inherited mutations in the X-linked gene CLCN4 are associated with syndromic intellectual disability and behavior and seizure disorders in males and females.Mol Psychiatry. 2018 Feb;23(2):222-230. doi: 10.1038/mp.2016.135. Epub 2016 Aug 23. Mol Psychiatry. 2018. PMID: 27550844 Free PMC article.
-
Inherited and acquired disorders of myelin: The underlying myelin pathology.Exp Neurol. 2016 Sep;283(Pt B):452-75. doi: 10.1016/j.expneurol.2016.04.002. Epub 2016 Apr 9. Exp Neurol. 2016. PMID: 27068622 Free PMC article. Review.
-
Drosophila ClC-a is required in glia of the stem cell niche for proper neurogenesis and wiring of neural circuits.Glia. 2019 Dec;67(12):2374-2398. doi: 10.1002/glia.23691. Epub 2019 Sep 3. Glia. 2019. PMID: 31479171 Free PMC article.
-
Glial regulators of ions and solutes required for specific chemosensory functions in Caenorhabditis elegans.iScience. 2022 Dec 1;25(12):105684. doi: 10.1016/j.isci.2022.105684. eCollection 2022 Dec 22. iScience. 2022. PMID: 36567707 Free PMC article.
-
Photoreceptor degeneration, azoospermia, leukoencephalopathy, and abnormal RPE cell function in mice expressing an early stop mutation in CLCN2.Invest Ophthalmol Vis Sci. 2010 Jun;51(6):3264-72. doi: 10.1167/iovs.09-4887. Epub 2010 Jan 13. Invest Ophthalmol Vis Sci. 2010. PMID: 20071672 Free PMC article.
References
-
- Bhat RV, Axt KJ, Fosnaugh JS, Smith KJ, Johnson KA, Hill DE, Kinzler KW, Baraban JM. Expression of the APC tumor suppressor protein in oligodendroglia. Glia. 1996;17:169–174. - PubMed
-
- Cid LP, Montrose-Rafizadeh C, Smith DI, Guggino WB, Cutting GR. Cloning of a putative human voltage-gated chloride channel (ClC-2) cDNA widely expressed in human tissues. Hum Mol Genet. 1995;4:407–413. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
