Phase 1 clinical trial of apical membrane antigen 1: an asexual blood-stage vaccine for Plasmodium falciparum malaria
- PMID: 15908397
- PMCID: PMC1111886
- DOI: 10.1128/IAI.73.6.3677-3685.2005
Phase 1 clinical trial of apical membrane antigen 1: an asexual blood-stage vaccine for Plasmodium falciparum malaria
Abstract
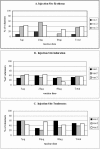
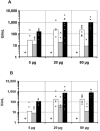
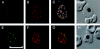
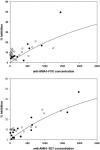
Apical membrane antigen 1 (AMA1), a polymorphic merozoite surface protein, is a leading blood-stage malaria vaccine candidate. A phase 1 trial was conducted with 30 malaria-naive volunteers to assess the safety and immunogenicity of the AMA1-C1 malaria vaccine. AMA1-C1 contains an equal mixture of recombinant proteins based on sequences from the FVO and 3D7 clones of Plasmodium falciparum. The proteins were expressed in Pichia pastoris and adsorbed on Alhydrogel. Ten volunteers in each of three dose groups (5 mug, 20 mug, and 80 mug) were vaccinated in an open-label study at 0, 28, and 180 days. The vaccine was well tolerated, with pain at the injection site being the most commonly observed reaction. Anti-AMA1 immunoglobulin G (IgG) was detected by enzyme-linked immunosorbent assay (ELISA) in 15/28 (54%) volunteers after the second immunization and in 23/25 (92%) after the third immunization, with equal reactivity to both AMA1-FVO and AMA1-3D7 vaccine components. A significant dose-response relationship between antigen dose and antibody response by ELISA was observed, and the antibodies were predominantly of the IgG1 isotype. Confocal microscopic evaluation of sera from vaccinated volunteers demonstrated reactivity with P. falciparum schizonts in a pattern similar to native parasite AMA1. Antigen-specific in vitro inhibition of both FVO and 3D7 parasites was achieved with IgG purified from sera of vaccinees, demonstrating biological activity of the antibodies. To our knowledge, this is the first AMA1 vaccine candidate to elicit functional immune responses in malaria-naive humans, and our results support the further development of this vaccine.
Figures
Similar articles
-
Phase 1 randomized controlled trial to evaluate the safety and immunogenicity of recombinant Pichia pastoris-expressed Plasmodium falciparum apical membrane antigen 1 (PfAMA1-FVO [25-545]) in healthy Malian adults in Bandiagara.Malar J. 2016 Aug 30;15(1):442. doi: 10.1186/s12936-016-1466-4. Malar J. 2016. PMID: 27577237 Free PMC article. Clinical Trial.
-
Phase I dose escalation safety and immunogenicity trial of Plasmodium falciparum apical membrane protein (AMA-1) FMP2.1, adjuvanted with AS02A, in malaria-naïve adults at the Walter Reed Army Institute of Research.Vaccine. 2007 May 22;25(21):4203-12. doi: 10.1016/j.vaccine.2007.03.012. Epub 2007 Mar 26. Vaccine. 2007. PMID: 17442466 Clinical Trial.
-
Safety and immunogenicity of a recombinant Plasmodium falciparum AMA1-DiCo malaria vaccine adjuvanted with GLA-SE or Alhydrogel® in European and African adults: A phase 1a/1b, randomized, double-blind multi-centre trial.Vaccine. 2017 Oct 27;35(45):6218-6227. doi: 10.1016/j.vaccine.2017.09.027. Epub 2017 Sep 22. Vaccine. 2017. PMID: 28947345 Clinical Trial.
-
Recombinant protein vaccines against the asexual blood stages of Plasmodium falciparum.Hum Vaccin. 2010 Jan;6(1):39-53. doi: 10.4161/hv.6.1.10712. Epub 2010 Jan 19. Hum Vaccin. 2010. PMID: 20061790 Review.
-
Malaria vaccine trials: the missing qualitative data.Immunol Cell Biol. 1996 Aug;74(4):301-5. doi: 10.1038/icb.1996.54. Immunol Cell Biol. 1996. PMID: 8872178 Review.
Cited by
-
Structure of the malaria antigen AMA1 in complex with a growth-inhibitory antibody.PLoS Pathog. 2007 Sep 7;3(9):1308-19. doi: 10.1371/journal.ppat.0030138. PLoS Pathog. 2007. PMID: 17907804 Free PMC article.
-
Enhanced antibody production in mice to the malaria antigen AMA1 by CPG 7909 requires physical association of CpG and antigen.Vaccine. 2007 Jul 20;25(29):5343-7. doi: 10.1016/j.vaccine.2007.05.007. Epub 2007 May 25. Vaccine. 2007. PMID: 17566616 Free PMC article.
-
Space-time clustering of childhood malaria at the household level: a dynamic cohort in a Mali village.BMC Public Health. 2006 Nov 21;6:286. doi: 10.1186/1471-2458-6-286. BMC Public Health. 2006. PMID: 17118176 Free PMC article.
-
Passive immunization with a multicomponent vaccine against conserved domains of apical membrane antigen 1 and 235-kilodalton rhoptry proteins protects mice against Plasmodium yoelii blood-stage challenge infection.Infect Immun. 2006 Oct;74(10):5529-36. doi: 10.1128/IAI.00573-06. Infect Immun. 2006. PMID: 16988228 Free PMC article.
-
Estimation on local transmission of malaria by serological approach under low transmission setting in Myanmar.Malar J. 2018 Jan 5;17(1):6. doi: 10.1186/s12936-017-2170-8. Malar J. 2018. PMID: 29304809 Free PMC article.
References
-
- Anonymous. 2000. Expert Committee on Malaria. WHO Tech. Rep. Ser. 892:i-v, 1-74. - PubMed
-
- Collins, W. E., D. Pye, P. E. Crewther, K. L. Vandenberg, G. G. Galland, A. J. Sulzer, D. J. Kemp, S. J. Edwards, R. L. Coppel, J. S. Sullivan, et al. 1994. Protective immunity induced in squirrel monkeys with recombinant apical membrane antigen-1 of Plasmodium fragile. Am. J. Trop. Med. Hyg. 51:711-719. - PubMed
-
- Deans, J. A., A. M. Knight, W. C. Jean, A. P. Waters, S. Cohen, and G. H. Mitchell. 1988. Vaccination trials in rhesus monkeys with a minor, invariant, Plasmodium knowlesi 66 kD merozoite antigen. Parasite Immunol. 10:535-552. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

