Wiskott-Aldrich syndrome protein is required for NK cell cytotoxicity and colocalizes with actin to NK cell-activating immunologic synapses
- PMID: 12177428
- PMCID: PMC123260
- DOI: 10.1073/pnas.162376099
Wiskott-Aldrich syndrome protein is required for NK cell cytotoxicity and colocalizes with actin to NK cell-activating immunologic synapses
Abstract
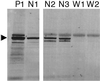
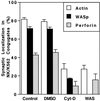
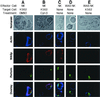
The Wiskott-Aldrich syndrome (WAS) is a primary immunodeficiency disorder caused by a mutation in WAS protein (WASp) that results in defective actin polymerization. Although the function of many hematopoietic cells requires WASp, the specific expression and function of this molecule in natural killer (NK) cells is unknown. Here, we report that WAS patients have increased percentages of peripheral blood NK cells and that fresh enriched NK cells from two patients with a WASp mutation have defective cytolytic function. In normal NK cells, WASp was expressed and localized to the activating immunologic synapse (IS) with filamentous actin (F-actin). Perforin also localized to the NK cell-activating IS but at a lesser frequency than F-actin and WASp. The accumulation of F-actin and WASp at the activating IS was decreased significantly in NK cells that had been treated with the inhibitor of actin polymerization, cytochalasin D. NK cells from WAS patients lacked expression of WASp and accumulated F-actin at the activating IS infrequently. Thus, WASp has an important function in NK cells. In patients with WASp mutations, the resulting NK cell defects are likely to contribute to their disease.
Figures


Similar articles
-
IL-2 induces a WAVE2-dependent pathway for actin reorganization that enables WASp-independent human NK cell function.J Clin Invest. 2011 Apr;121(4):1535-48. doi: 10.1172/JCI44862. Epub 2011 Mar 7. J Clin Invest. 2011. PMID: 21383498 Free PMC article.
-
The Wiskott-Aldrich syndrome protein regulates nuclear translocation of NFAT2 and NF-kappa B (RelA) independently of its role in filamentous actin polymerization and actin cytoskeletal rearrangement.J Immunol. 2005 Mar 1;174(5):2602-11. doi: 10.4049/jimmunol.174.5.2602. J Immunol. 2005. PMID: 15728466
-
Impaired natural and CD16-mediated NK cell cytotoxicity in patients with WAS and XLT: ability of IL-2 to correct NK cell functional defect.Blood. 2004 Jul 15;104(2):436-43. doi: 10.1182/blood-2003-07-2621. Epub 2004 Mar 4. Blood. 2004. PMID: 15001467
-
[Molecular pathogenesis of Wiskott-Aldrich syndrome].Nihon Rinsho Meneki Gakkai Kaishi. 2005 Jun;28(3):140-7. doi: 10.2177/jsci.28.140. Nihon Rinsho Meneki Gakkai Kaishi. 2005. PMID: 15997177 Review. Japanese.
-
Wiskott-Aldrich Syndrome: a model for defective actin reorganization, cell trafficking and synapse formation.Curr Opin Immunol. 2003 Oct;15(5):585-91. doi: 10.1016/s0952-7915(03)00112-2. Curr Opin Immunol. 2003. PMID: 14499269 Review.
Cited by
-
WIP remodeling actin behind the scenes: how WIP reshapes immune and other functions.Int J Mol Sci. 2012;13(6):7629-7647. doi: 10.3390/ijms13067629. Epub 2012 Jun 21. Int J Mol Sci. 2012. PMID: 22837718 Free PMC article. Review.
-
Natural killer cell cytotoxicity: how do they pull the trigger?Immunology. 2009 Sep;128(1):7-15. doi: 10.1111/j.1365-2567.2009.03123.x. Immunology. 2009. PMID: 19689731 Free PMC article. Review.
-
Signal transduction during activation and inhibition of natural killer cells.Curr Protoc Immunol. 2010 Aug;Chapter 11:Unit 11.9B. doi: 10.1002/0471142735.im1109bs90. Curr Protoc Immunol. 2010. PMID: 20814939 Free PMC article. Review.
-
Impaired NK-cell migration in WAS/XLT patients: role of Cdc42/WASp pathway in the control of chemokine-induced beta2 integrin high-affinity state.Blood. 2010 Apr 8;115(14):2818-26. doi: 10.1182/blood-2009-07-235804. Epub 2010 Feb 3. Blood. 2010. PMID: 20130240 Free PMC article.
-
Actin Retrograde Flow Regulated by the Wiskott-Aldrich Syndrome Protein Drives the Natural Killer Cell Response.Cancers (Basel). 2022 Aug 2;14(15):3756. doi: 10.3390/cancers14153756. Cancers (Basel). 2022. PMID: 35954420 Free PMC article.
References
-
- Derry J. M., Ochs, H. D. & Francke, U. (1994) Cell 78, 635-644. - PubMed
-
- Higgs H. N. & Pollard, T. D. (2001) Annu. Rev. Biochem. 70, 649-676. - PubMed
-
- Thrasher A. J., Burns, S., Lorenzi, R. & Jones, G. E. (2000) Immunol. Rev. 178, 118-128. - PubMed
-
- Sullivan K. E., Mullen, C. A., Blaese, R. M. & Winkelstein, J. A. (1994) J. Pediatr. 125, 876-885. - PubMed
-
- Miller J. S. (2001) Exp. Hematol. 29, 1157-1168. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

