Comparative analysis of integrin expression on monocyte-derived macrophages and monocyte-derived dendritic cells
- PMID: 10929059
- PMCID: PMC2327027
- DOI: 10.1046/j.1365-2567.2000.00056.x
Comparative analysis of integrin expression on monocyte-derived macrophages and monocyte-derived dendritic cells
Abstract
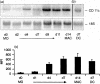
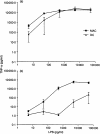
Both macrophages (MAC) and dendritic cells (DC) are members of the mononuclear phagocyte system (MPS) with monocytes (MO) as common precursor cells. Cells of the MPS are able to take up, process and present antigens to T lymphocytes, thereby inducing a primary or secondary immune response. Adhesion molecules are of crucial importance for the interaction of antigen-presenting cells with immune cells, especially T lymphocytes. By representational difference analysis, we identified CD49c (VLA-3), a member of the beta1-integrin family of adhesion receptors, as differentiation-associated antigen in MO-derived MAC. In contrast, MO-derived DC did not express CD49c mRNA. These data prompted us to compare the integrin expression pattern of MAC and DC. Both cell types showed a low expression of the alpha-chains of the beta1-integrins CD49a, CD49b, CD49d and CD49e, whereas a marked difference was observed for CD49c and CD49f. Expression of both integrins increased during MO to MAC differentiation, but was not detectable on DC. In parallel the beta1-chain (CD29) was clearly up-regulated during MO to MAC differentiation but was only weakly expressed on DC. On the other hand, the beta2-integrins CD11a, CD11b, CD11c and CD18 were all expressed on MAC and DC. Beside their role in cell-cell interaction and adhesion, beta2-integrins are also known as possible binding molecules for bacteria and lipopolysaccharide (LPS), especially for high LPS concentrations. Therefore we investigated the LPS response of MAC versus DC in terms of tumour necrosis factor-alpha (TNF-alpha) release. DC were less responsive to low doses of LPS, which can easily be explained by the very low CD14 expression on DC compared for MAC. In contrast, the TNF-alpha response was comparable to MAC when DC were stimulated with high LPS concentrations. Our results show a specific, differentiation-dependent pattern of beta1- and beta2-integrin expression on in vitro-generated MAC and DC. We suggest that the high expression of CD11/CD18 on DC could be involved in the LPS binding of DC. As LPS is not only an activation but also a differentiation stimulus for DC, the expression of CD11/CD18 on DC may be important for the successful maturation of DC and thereby the initiation of a primary immune response.
Figures

Similar articles
-
Inverse regulation of the ADAM-family members, decysin and MADDAM/ADAM19 during monocyte differentiation.Immunology. 2003 Dec;110(4):450-7. doi: 10.1111/j.1365-2567.2003.01754.x. Immunology. 2003. PMID: 14632642 Free PMC article.
-
Integrin regulation of leukocyte inflammatory functions. CD11b/CD18 enhancement of the tumor necrosis factor-alpha responses of monocytes.J Immunol. 1993 Apr 1;150(7):2972-80. J Immunol. 1993. PMID: 8095957
-
Maturation-dependent expression and function of the CD49d integrin on monocyte-derived human dendritic cells.J Immunol. 2000 Oct 15;165(8):4338-45. doi: 10.4049/jimmunol.165.8.4338. J Immunol. 2000. PMID: 11035069
-
Myb-transformed hematopoietic cells as a model for monocyte differentiation into dendritic cells and macrophages.J Leukoc Biol. 1999 Aug;66(2):217-23. doi: 10.1002/jlb.66.2.217. J Leukoc Biol. 1999. PMID: 10449157 Review.
-
β2 Integrins As Regulators of Dendritic Cell, Monocyte, and Macrophage Function.Front Immunol. 2017 Dec 20;8:1866. doi: 10.3389/fimmu.2017.01866. eCollection 2017. Front Immunol. 2017. PMID: 29326724 Free PMC article. Review.
Cited by
-
HIV-1 tat promotes integrin-mediated HIV transmission to dendritic cells by binding Env spikes and competes neutralization by anti-HIV antibodies.PLoS One. 2012;7(11):e48781. doi: 10.1371/journal.pone.0048781. Epub 2012 Nov 13. PLoS One. 2012. PMID: 23152803 Free PMC article.
-
Circulating classical monocytes are associated with CD11c+ macrophages in human visceral adipose tissue.Sci Rep. 2017 Feb 15;7:42665. doi: 10.1038/srep42665. Sci Rep. 2017. PMID: 28198418 Free PMC article.
-
RGD-Modified Albumin Nanoconjugates for Targeted Delivery of a Porphyrin Photosensitizer.Mol Pharm. 2017 Aug 7;14(8):2793-2804. doi: 10.1021/acs.molpharmaceut.7b00321. Epub 2017 Jul 25. Mol Pharm. 2017. PMID: 28700237 Free PMC article.
-
Monocyte Adhesion, Migration, and Extracellular Matrix Breakdown Are Regulated by Integrin αVβ3 in Mycobacterium tuberculosis Infection.J Immunol. 2017 Aug 1;199(3):982-991. doi: 10.4049/jimmunol.1700128. Epub 2017 Jun 23. J Immunol. 2017. PMID: 28646039 Free PMC article.
-
Microparticle surface modifications targeting dendritic cells for non-activating applications.Biomaterials. 2012 Oct;33(29):7221-32. doi: 10.1016/j.biomaterials.2012.06.049. Epub 2012 Jul 12. Biomaterials. 2012. PMID: 22796161 Free PMC article.
References
-
- Andreesen R, Picht J, Lohr GW. Primary cultures of human blood-born macrophages grown on hydrophobic teflon membranes. J Immunol Methods. 1983;56:295. - PubMed
-
- Romani N, Reider D, Heuer M, et al. Generation of mature dendritic cells from human blood. An improved method with special regard to clinical applicability. J Immunol Methods. 1996;196:137. - PubMed
-
- Hart DN. Dendritic cells: unique leukocyte populations which control the primary immune response. Blood. 1997;90:3245. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

