Expression of reelin in adult mammalian blood, liver, pituitary pars intermedia, and adrenal chromaffin cells
- PMID: 10655522
- PMCID: PMC15597
- DOI: 10.1073/pnas.97.3.1281
Expression of reelin in adult mammalian blood, liver, pituitary pars intermedia, and adrenal chromaffin cells
Abstract
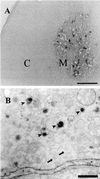
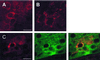
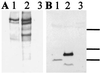
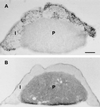
Reelin regulates telencephalic and cerebellar lamination during mammalian development and is expressed in several structures of the adult brain; however, only traces of reelin were believed to be in peripheral tissues. Because reelin structurally resembles extracellular matrix proteins, and because many of these proteins are expressed in blood, we hypothesized that reelin also might be detectable in the circulation. Reelin (420 kDa) and two reelin-like immunoreactive bands (310 and 160 kDa) are expressed in serum and platelet-poor plasma of rats, mice, and humans, but these three bands were not detectable in serum of homozygous reeler (rl/rl) mice. Reelin plasma levels in heterozygous (rl/+) mice were half of those in wild-type littermates. Western blotting and immunocytochemistry using antireelin mAbs indicated that reelin-like immunoreactivity was expressed in a subset of chromaffin cells within the rat adrenal medulla and in a subset of cells coexpressing alpha-melanocyte-stimulating hormone within the pituitary pars intermedia. However, surgical removal of adrenal or pituitary failed to decrease the amount of reelin (420-kDa band) expressed in serum. Adult liver expressed one-third of the reelin mRNA concentration expressed in adult mouse cerebral cortex. Full-length reelin protein was detectable in liver extracts in situ; acutely isolated liver cells also secreted full-length reelin in vitro. Liver appears to be a prime candidate to produce and maintain the circulating reelin pool. It now becomes relevant to ask whether circulating reelin has a physiologic role on one or more peripheral target tissues.
Figures

Similar articles
-
Reelin in the extracellular matrix and dendritic spines of the cortex and hippocampus: a comparison between wild type and heterozygous reeler mice by immunoelectron microscopy.J Neurocytol. 2001 May;30(5):413-25. doi: 10.1023/a:1015017710332. J Neurocytol. 2001. PMID: 11951052
-
A truncated Reelin protein is produced but not secreted in the 'Orleans' reeler mutation (Reln[rl-Orl]).Brain Res Mol Brain Res. 1997 Oct 15;50(1-2):85-90. doi: 10.1016/s0169-328x(97)00166-6. Brain Res Mol Brain Res. 1997. PMID: 9406921
-
Expression of reelin, the gene responsible for the reeler mutation, in embryonic development and adulthood in the mouse.Dev Dyn. 1997 Oct;210(2):157-72. doi: 10.1002/(SICI)1097-0177(199710)210:2<157::AID-AJA8>3.0.CO;2-F. Dev Dyn. 1997. PMID: 9337136
-
Role of reelin in the control of brain development.Brain Res Brain Res Rev. 1998 May;26(2-3):285-94. doi: 10.1016/s0165-0173(97)00035-0. Brain Res Brain Res Rev. 1998. PMID: 9651544 Review.
-
[Cytoarchitectonic abnormality in the facial nucleus of the reeler mouse].Kaibogaku Zasshi. 1999 Aug;74(4):411-20. Kaibogaku Zasshi. 1999. PMID: 10496086 Review. Japanese.
Cited by
-
Clinical and Neurobiological Relevance of Current Animal Models of Autism Spectrum Disorders.Biomol Ther (Seoul). 2016 May 1;24(3):207-43. doi: 10.4062/biomolther.2016.061. Biomol Ther (Seoul). 2016. PMID: 27133257 Free PMC article. Review.
-
Dissecting bipolar disorder complexity through epigenomic approach.Mol Psychiatry. 2016 Nov;21(11):1490-1498. doi: 10.1038/mp.2016.123. Epub 2016 Aug 2. Mol Psychiatry. 2016. PMID: 27480490 Free PMC article. Review.
-
Reelin expression and glycosylation patterns are altered in Alzheimer's disease.Proc Natl Acad Sci U S A. 2006 Apr 4;103(14):5573-8. doi: 10.1073/pnas.0601279103. Epub 2006 Mar 27. Proc Natl Acad Sci U S A. 2006. PMID: 16567613 Free PMC article.
-
Methodological factors influencing measurement and processing of plasma reelin in humans.BMC Biochem. 2003 Sep 7;4:9. doi: 10.1186/1471-2091-4-9. BMC Biochem. 2003. PMID: 12959647 Free PMC article.
-
Reelin: Neurodevelopmental Architect and Homeostatic Regulator of Excitatory Synapses.J Biol Chem. 2017 Jan 27;292(4):1330-1338. doi: 10.1074/jbc.R116.766782. Epub 2016 Dec 19. J Biol Chem. 2017. PMID: 27994051 Free PMC article. Review.
References
-
- Rakic P, Caviness V S. Neuron. 1995;14:1101–1104. - PubMed
-
- Lambert de Rouvroit C, Goffinet A M. Adv Anat Embryol Cell Biol. 1998;150:1–108. - PubMed
-
- D'Arcangelo G, Miao G G, Chen S-C, Soares H D, Morgan J I, Curran T. Nature (London) 1995;374:719–723. - PubMed
-
- Ogawa M, Miyata T, Nakajima K, Yagyu K, Seike M, Ikenaka K, Yamamoto H, Mikoshiba K. Neuron. 1995;14:899–912. - PubMed
-
- Hirotsune S, Takahara T, Sasaki N, Hirose K, Yoshiki A, Ohashi T, Kusakabe M, Murakami Y, Muramatsu M, Watanabe S, et al. Nat Genet. 1995;10:77–83. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

