Current practices and challenges in adaptation of clinical guidelines: a qualitative study based on semistructured interviews
- PMID: 34857574
- PMCID: PMC8640632
- DOI: 10.1136/bmjopen-2021-053587
Current practices and challenges in adaptation of clinical guidelines: a qualitative study based on semistructured interviews
Abstract
Objective: This study aims to better understand the current practice of clinical guideline adaptation and identify challenges raised in this process, given that published adapted clinical guidelines are generally of low quality, poorly reported and not based on published frameworks.
Design: A qualitative study based on semistructured interviews. We conducted a framework analysis for the adaptation process, and thematic analysis for participants' views and experiences about adaptation process.
Setting: Nine guideline development organisations from seven countries.
Participants: Guideline developers who have adapted clinical guidelines within the last 3 years. We identified potential participants through published adapted clinical guidelines, recommendations from experts, and a review of the Guideline International Network Conference attendees' list.
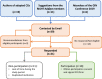
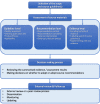
Results: We conducted ten interviews and identified nine adaptation methodologies. The reasons for adapting clinical guidelines include developing de novo clinical guidelines, implementing source clinical guidelines, and harmonising and updating existing clinical guidelines. We identified the following core steps of the adaptation process (1) selection of scope and source guideline(s), (2) assessment of source materials (guidelines, recommendations and evidence level), (3) decision-making process and (4) external review and follow-up process. Challenges on the adaptation of clinical guidelines include limitations from source clinical guidelines (poor quality or reporting), limitations from adaptation settings (lacking resources or skills), adaptation process intensity and complexity, and implementation barriers. We also described how participants address the complexities and implementation issues of the adaptation process.
Conclusions: Adaptation processes have been increasingly used to develop clinical guidelines, with the emergence of different purposes. The identification of core steps and assessment levels could help guideline adaptation developers streamline their processes. More methodological research is needed to develop rigorous international standards for adapting clinical guidelines.
Keywords: qualitative research; quality in health care; statistics & research methods.
© Author(s) (or their employer(s)) 2021. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: EAA has intellectual CoIs related to his contribution to the development of methods of guideline adaptation, the RIGHT statement and methodological studies in the field.
Figures
Similar articles
-
The future of Cochrane Neonatal.Early Hum Dev. 2020 Nov;150:105191. doi: 10.1016/j.earlhumdev.2020.105191. Epub 2020 Sep 12. Early Hum Dev. 2020. PMID: 33036834
-
Variations in processes for guideline adaptation: a qualitative study of World Health Organization staff experiences in implementing guidelines.BMC Public Health. 2020 Nov 23;20(1):1758. doi: 10.1186/s12889-020-09812-0. BMC Public Health. 2020. PMID: 33228608 Free PMC article.
-
Barriers and facilitators in developing patient versions of clinical practice guidelines - qualitative interviews on experiences of international guideline producers.BMC Health Serv Res. 2024 Jan 16;24(1):78. doi: 10.1186/s12913-023-10524-5. BMC Health Serv Res. 2024. PMID: 38229078 Free PMC article.
-
Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome.Fertil Steril. 2018 Aug;110(3):364-379. doi: 10.1016/j.fertnstert.2018.05.004. Epub 2018 Jul 19. Fertil Steril. 2018. PMID: 30033227 Free PMC article. Review.
-
Adaptation, evaluation, and updating of guidelines: article 14 in Integrating and coordinating efforts in COPD guideline development. An official ATS/ERS workshop report.Proc Am Thorac Soc. 2012 Dec;9(5):304-10. doi: 10.1513/pats.201208-067ST. Proc Am Thorac Soc. 2012. PMID: 23256175 Review.
Cited by
-
Strengthening guideline contextualization in the WHO European Region.Bull World Health Organ. 2024 Oct 1;102(10):715-721. doi: 10.2471/BLT.24.291779. Epub 2024 Aug 23. Bull World Health Organ. 2024. PMID: 39318892 Free PMC article.
-
Translating guidelines into practice via implementation science: an update in lipidology.Curr Opin Lipidol. 2022 Dec 1;33(6):336-341. doi: 10.1097/MOL.0000000000000835. Epub 2022 Jun 27. Curr Opin Lipidol. 2022. PMID: 35779062 Free PMC article. Review.
-
Determinants of regulatory compliance in health and social care services: A systematic review using the Consolidated Framework for Implementation Research.PLoS One. 2023 Apr 13;18(4):e0278007. doi: 10.1371/journal.pone.0278007. eCollection 2023. PLoS One. 2023. PMID: 37053186 Free PMC article.
-
Development and pilot testing of the Population And ContExt adaption of decision aids (PACE) framework.PEC Innov. 2024 Sep 30;5:100347. doi: 10.1016/j.pecinn.2024.100347. eCollection 2024 Dec 15. PEC Innov. 2024. PMID: 39430917 Free PMC article.
References
-
- Okely AD, Ghersi D, Hesketh KD, et al. . A collaborative approach to adopting/adapting guidelines - The Australian 24-Hour Movement Guidelines for the early years (Birth to 5 years): an integration of physical activity, sedentary behavior, and sleep. BMC Public Health 2017;17:869. 10.1186/s12889-017-4867-6 - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
