Fluorochrome-linked immunoassay for functional analysis of the mannose binding lectin complement pathway to the level of C3 cleavage
- PMID: 17512534
- PMCID: PMC1976379
- DOI: 10.1016/j.jim.2007.04.004
Fluorochrome-linked immunoassay for functional analysis of the mannose binding lectin complement pathway to the level of C3 cleavage
Abstract
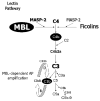
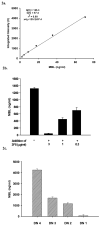
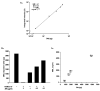
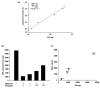
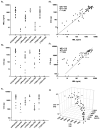
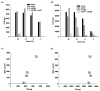
The humoral response to invading pathogens is mediated by a repertoire of innate immune molecules and receptors able to recognize pathogen-associated molecular patterns. Mannose binding lectin (MBL) and ficolins are initiation molecules of the lectin complement pathway (LCP) that bridge innate and adaptive immunity. Activation of the MBL-dependent lectin pathway, to the level of C3 cleavage, requires functional MASP-2, C2, C4 and C3, all of which have been identified with genetic polymorphisms that can affect protein concentration and function. Current assays for MBL and MASP-2 lack the ability to assess activation of all components to the level of C3 cleavage in a single assay platform. We developed a novel, low volume, fluorochrome linked immunoassay (FLISA) that quantitatively assesses the functional status of MBL, MASP-2 and C3 convertase in a single well. The assay can be used with plasma or serum. Multiple freeze/thaw cycles of serum do not significantly alter the assay, making it ideal for high throughput of large sample databases with minimal volume use. The FLISA can be used potentially to identify specific human disease correlations between these components and clinical outcomes in already established databases.
Figures
Similar articles
-
Structural and functional overview of the lectin complement pathway: its molecular basis and physiological implication.Arch Immunol Ther Exp (Warsz). 2013 Aug;61(4):273-83. doi: 10.1007/s00005-013-0229-y. Epub 2013 Apr 7. Arch Immunol Ther Exp (Warsz). 2013. PMID: 23563865 Review.
-
Co-complexes of MASP-1 and MASP-2 associated with the soluble pattern-recognition molecules drive lectin pathway activation in a manner inhibitable by MAp44.J Immunol. 2013 Aug 1;191(3):1334-45. doi: 10.4049/jimmunol.1300780. Epub 2013 Jun 19. J Immunol. 2013. PMID: 23785123
-
Cooperation between MASP-1 and MASP-2 in the generation of C3 convertase through the MBL pathway.Int Immunol. 2007 Feb;19(2):141-9. doi: 10.1093/intimm/dxl131. Epub 2006 Dec 20. Int Immunol. 2007. PMID: 17182967
-
Essential Roles for Mannose-Binding Lectin-Associated Serine Protease-1/3 in the Development of Lupus-Like Glomerulonephritis in MRL/lpr Mice.Front Immunol. 2018 May 28;9:1191. doi: 10.3389/fimmu.2018.01191. eCollection 2018. Front Immunol. 2018. PMID: 29892304 Free PMC article.
-
Toward a structure-based comprehension of the lectin pathway of complement.Mol Immunol. 2013 Dec;56(4):413-22. doi: 10.1016/j.molimm.2013.05.007. Epub 2013 Aug 1. Mol Immunol. 2013. PMID: 23911397 Review.
Cited by
-
Complement inhibition prevents oncolytic vaccinia virus neutralization in immune humans and cynomolgus macaques.Mol Ther. 2015 Jun;23(6):1066-1076. doi: 10.1038/mt.2015.49. Epub 2015 Mar 25. Mol Ther. 2015. PMID: 25807289 Free PMC article.
-
Circulating mannan-binding lectin, M-, L-, H-ficolin and collectin-liver-1 levels in patients with acute liver failure.Liver Int. 2015 Mar;35(3):756-63. doi: 10.1111/liv.12682. Epub 2014 Sep 30. Liver Int. 2015. PMID: 25203057 Free PMC article.
-
Complement activation and cardiac surgery: a novel target for improving outcomes.Anesth Analg. 2012 Oct;115(4):759-71. doi: 10.1213/ANE.0b013e3182652b7d. Epub 2012 Jul 13. Anesth Analg. 2012. PMID: 22798530 Free PMC article. Review.
-
The mannose-binding lectin pathway is a significant contributor to reperfusion injury in the type 2 diabetic heart.Diab Vasc Dis Res. 2009 Jul;6(3):172-80. doi: 10.1177/1479164109336051. Diab Vasc Dis Res. 2009. PMID: 20216929 Free PMC article.
-
Mannose-binding lectin-associated serine protease-1 is a significant contributor to coagulation in a murine model of occlusive thrombosis.J Immunol. 2012 Jan 15;188(2):885-91. doi: 10.4049/jimmunol.1102916. Epub 2011 Dec 12. J Immunol. 2012. PMID: 22156595 Free PMC article.
References
-
- Baccarelli A, Hou L, Chen J, Lissowska J, El-Omar EM, Grillo P, Giacomini SM, Yaeger M, Bernig T, Zatonski W, Fraumeni JF, Jr, Chanock SJ, Chow WH. Mannose-binding lectin-2 genetic variation and stomach cancer risk. Int J Cancer. 2006;119:1970. - PubMed
-
- Bathum L, Hansen H, Teisner B, Koch C, Garred P, Rasmussen K, Wang P. Association between combined properdin and mannose-binding lectin deficiency and infection with Neisseria meningitidis. Mol Immunol. 2006;43:473. - PubMed
-
- Bax WA, Cluysenaer OJ, Bartelink AK, Aerts PC, Ezekowitz RA, van DH. Association of familial deficiency of mannose-binding lectin and meningococcal disease. Lancet. 1999;354:1094. - PubMed
-
- Best LG, Davidson M, North KE, MacCluer JW, Zhang Y, Lee ET, Howard BV, DeCroo S, Ferrell RE. Prospective analysis of mannose-binding lectin genotypes and coronary artery disease in American Indians: the Strong Heart Study. Circ. 2004;109:471. - PubMed
-
- Bouwman LH, Eerligh P, Terpstra OT, Daha MR, de KP, Ballieux BE, Bruining GJ, Van Der Slik AR, Roos A, Roep BO. Elevated levels of mannose-binding lectin at clinical manifestation of type 1 diabetes in juveniles. Diabetes. 2005;54:3002. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

