Zolbetuximab for Unresectable and Metastatic Gastric and Gastroesophageal Junction Adenocarcinoma: A Review of Literature
- PMID: 39759684
- PMCID: PMC11700525
- DOI: 10.7759/cureus.75206
Zolbetuximab for Unresectable and Metastatic Gastric and Gastroesophageal Junction Adenocarcinoma: A Review of Literature
Abstract
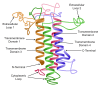
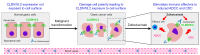
This article comprehensively reviews the working, efficacy, and safety profile of zolbetuximab. Zolbetuximab is a pioneering chimeric monoclonal antibody designed to target Claudin 18.2 (CLDN18.2), a tight junction protein frequently overexpressed in various gastrointestinal cancers, including gastric (G) and gastroesophageal junction (GEJ) adenocarcinomas. This drug has captured attention in the treatment of unresectable and metastatic G/GEJ cancer, especially in HER2-negative patients whose tumours express CLDN18.2. Zolbetuximab is a drug that binds to CLDN18.2, and its binding initiates an immune response that attacks and kills the cancer cells. It is typically co-prescribed with fluoropyrimidine and platinum-containing chemotherapy. The drug (formerly IMAB362), brand name Vyloy, was developed by Astellas Pharma, Tokyo, Japan. After multiple rounds of clinical trials, it was approved by the U.S. Food and Drug Administration (FDA) as a first-line treatment for locally advanced, unresectable cancer, establishing itself as a promising option for advanced G/GEJ cancers. Studies reveal that it targets CLDN18.2 for the selective killing of cancer cells while sparing most healthy tissues. Zolbetuximab has demonstrated a significant improvement in progression-free survival (PFS) when combined with chemotherapy (like mFOLFOX6 or CAPOX). Also, it showed prolonged PFS compared to chemotherapy alone in previously untreated, CLDN18.2-positive, HER2-negative patients. Zolbetuximab has also shown improvements in overall survival (OS), regardless of whether the cancer progresses. This is a crucial benefit for patients with advanced G/GEJ adenocarcinomas. Additionally, zolbetuximab's dual action triggers antibody-dependent cellular cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC). This enhances the immune system's ability to destroy cancerous cells and results in highly potent tumour destruction compared with chemotherapy alone. Zolbetuximab is a relatively safe drug with a few adverse effects. The most frequently reported side effects were gastrointestinal, namely nausea, fatigue, vomiting, decreased appetite, diarrhea, neutropenia, anemia, and thrombocytopenia, potentially due to specific CLDN18.2 expression in the gastric mucosa. Its side effects are generally manageable, with no unexpected toxicity beyond those typically seen in patients receiving chemotherapy.
Keywords: chemotherapy; cldn18.2-positive; gastric cancer; gastroesophageal junction adenocarcinoma; her2-negative; immunohistochemistry; monoclonal antibody; zolbetuximab-clzb.
Copyright © 2024, Samanta et al.
Conflict of interest statement
Conflicts of interest: In compliance with the ICMJE uniform disclosure form, all authors declare the following: Payment/services info: All authors have declared that no financial support was received from any organization for the submitted work. Financial relationships: All authors have declared that they have no financial relationships at present or within the previous three years with any organizations that might have an interest in the submitted work. Other relationships: All authors have declared that there are no other relationships or activities that could appear to have influenced the submitted work.
Figures
Similar articles
-
Zolbetuximab plus chemotherapy for locally advanced unresectable or metastatic stomach or gastroesophageal junction cancers: a plain language summary.Future Oncol. 2024;20(26):1861-1877. doi: 10.1080/14796694.2024.2342107. Epub 2024 May 24. Future Oncol. 2024. PMID: 38861294 Free PMC article.
-
First-line treatment with zolbetuximab plus CAPOX for ClDN18.2-positive gastric or gastroesophageal junction adenocarcinoma: a cost-effectiveness analysis.Therap Adv Gastroenterol. 2024 Nov 22;17:17562848241297052. doi: 10.1177/17562848241297052. eCollection 2024. Therap Adv Gastroenterol. 2024. PMID: 39582898 Free PMC article.
-
[A New Molecular Targeted Agent for Gastric Cancer-The Anti-Claudin 18.2 Antibody, Zolbetuximab].Gan To Kagaku Ryoho. 2024 Nov;51(11):1111-1118. Gan To Kagaku Ryoho. 2024. PMID: 39648123 Review. Japanese.
-
Far Posterior Approach for Rib Fracture Fixation: Surgical Technique and Tips.JBJS Essent Surg Tech. 2024 Dec 6;14(4):e23.00094. doi: 10.2106/JBJS.ST.23.00094. eCollection 2024 Oct-Dec. JBJS Essent Surg Tech. 2024. PMID: 39650795 Free PMC article.
-
Depressing time: Waiting, melancholia, and the psychoanalytic practice of care.In: Kirtsoglou E, Simpson B, editors. The Time of Anthropology: Studies of Contemporary Chronopolitics. Abingdon: Routledge; 2020. Chapter 5. In: Kirtsoglou E, Simpson B, editors. The Time of Anthropology: Studies of Contemporary Chronopolitics. Abingdon: Routledge; 2020. Chapter 5. PMID: 36137063 Free Books & Documents. Review.
References
-
- The preclinical discovery and development of zolbetuximab for the treatment of gastric cancer. Zeng Y, Lockhart AC, Jin RU. Expert Opin Drug Discov. 2024;19:873–886. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
