6-Gingerol Inhibits De Novo Lipogenesis by Targeting Stearoyl-CoA Desaturase to Alleviate Fructose-Induced Hepatic Steatosis
- PMID: 39457074
- PMCID: PMC11508832
- DOI: 10.3390/ijms252011289
6-Gingerol Inhibits De Novo Lipogenesis by Targeting Stearoyl-CoA Desaturase to Alleviate Fructose-Induced Hepatic Steatosis
Abstract
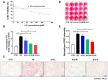
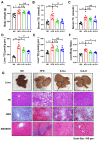
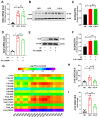
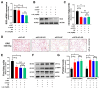
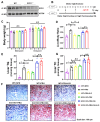
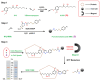
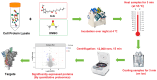
Metabolic-associated fatty liver disease (MAFLD), also known as non-alcoholic fatty liver disease (NAFLD), is a worldwide liver disease without definitive or widely used therapeutic drugs in clinical practice. In this study, we confirm that 6-gingerol (6-G), an active ingredient of ginger (Zingiber officinale Roscoe) in traditional Chinese medicine (TCM), can alleviate fructose-induced hepatic steatosis. It was found that 6-G significantly decreased hyperlipidemia caused by high-fructose diets (HFD) in rats, and reversed the increase in hepatic de novo lipogenesis (DNL) and triglyceride (TG) levels induced by HFD, both in vivo and in vitro. Mechanistically, chemical proteomics and cellular thermal shift assay (CETSA)-proteomics approaches revealed that stearoyl-CoA desaturase (SCD) is a direct binding target of 6-G, which was confirmed by further CETSA assay and molecular docking. Meanwhile, it was found that 6-G could not alter SCD expression (in either mRNA or protein levels), but inhibited SCD activity (decreasing the desaturation levels of fatty acids) in HFD-fed rats. Furthermore, SCD deficiency mimicked the ability of 6-G to reduce lipid accumulation in HF-induced HepG2 cells, and impaired the improvement in hepatic steatosis brought about by 6-G treatment in HFD supplemented with oleic acid diet-induced SCD1 knockout mice. Taken together, our present study demonstrated that 6-G inhibits DNL by targeting SCD to alleviate fructose diet-induced hepatic steatosis.
Keywords: 6-gingerol; hepatic steatosis; metabolic-associated fatty liver disease; stearoyl-CoA desaturase; target.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Long-term fructose consumption prolongs hepatic stearoyl-CoA desaturase 1 activity independent of upstream regulation in rats.Biochem Biophys Res Commun. 2016 Oct 28;479(4):643-648. doi: 10.1016/j.bbrc.2016.09.160. Epub 2016 Sep 30. Biochem Biophys Res Commun. 2016. PMID: 27697525
-
[6]-gingerol dampens hepatic steatosis and inflammation in experimental nonalcoholic steatohepatitis.Phytomedicine. 2015 Apr 15;22(4):452-61. doi: 10.1016/j.phymed.2015.01.015. Epub 2015 Feb 26. Phytomedicine. 2015. PMID: 25925967
-
Chronic administration of recombinant IL-6 upregulates lipogenic enzyme expression and aggravates high-fat-diet-induced steatosis in IL-6-deficient mice.Dis Model Mech. 2015 Jul 1;8(7):721-31. doi: 10.1242/dmm.019166. Epub 2015 May 14. Dis Model Mech. 2015. PMID: 26035386 Free PMC article.
-
Role of Dietary Fructose and Hepatic De Novo Lipogenesis in Fatty Liver Disease.Dig Dis Sci. 2016 May;61(5):1282-93. doi: 10.1007/s10620-016-4054-0. Epub 2016 Feb 8. Dig Dis Sci. 2016. PMID: 26856717 Free PMC article. Review.
-
Fructose as a key player in the development of fatty liver disease.World J Gastroenterol. 2013 Feb 28;19(8):1166-72. doi: 10.3748/wjg.v19.i8.1166. World J Gastroenterol. 2013. PMID: 23482247 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

