Arthropod prey type drives decomposition rates and microbial community processes
- PMID: 38916291
- PMCID: PMC11267907
- DOI: 10.1128/aem.00394-24
Arthropod prey type drives decomposition rates and microbial community processes
Abstract
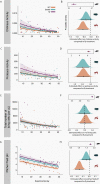
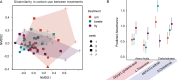
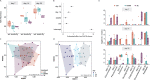
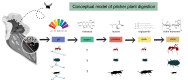
Microbial communities perform various functions, many of which contribute to ecosystem-level nutrient cycling via decomposition. Factors influencing leaf detrital decomposition are well understood in terrestrial and aquatic ecosystems, but much less is known about arthropod detrital inputs. Here, we sought to infer how differences in arthropod detritus affect microbial-driven decomposition and community function in a carnivorous pitcher plant, Sarracenia purpurea. Using sterile mesh bags filled with different types of sterile arthropod prey, we assessed if prey type influenced the rate of decomposition in pitcher plants over 7 weeks. Additionally, we measured microbial community composition and function, including hydrolytic enzyme activity and carbon substrate use. When comparing decomposition rates, we found that ant and beetle prey with higher exoskeleton content lost less mass compared with fly prey. We observed the highest protease activity in the fly treatment, which had the lowest exoskeleton content. Additionally, we saw differences in the pH of the pitcher fluid, driven by the ant treatment which had the lowest pH. According to our results from 16S rRNA gene metabarcoding, prey treatments with the highest bacterial amplicon sequence variant (ASV) richness (ant and beetle) were associated with prey that lost a lower proportion of mass over the 7 weeks. Overall, arthropod detritus provides unique nutrient sources to decomposer communities, with different prey influencing microbial hydrolytic enzyme activity and composition.
Importance: Microbial communities play pivotal roles in nutrient cycling via decomposition and nutrient transformation; however, it is often unclear how different substrates influence microbial activity and community composition. Our study highlights how different types of insects influence decomposition and, in turn, microbial composition and function. We use the aquatic pools found in a carnivorous pitcher plant as small, discrete ecosystems that we can manipulate and study independently. We find that some insect prey (flies) breaks down faster than others (beetles or ants) likely because flies contain more things that are easy for microbes to eat and derive essential nutrients from. This is also reflected in higher enzyme activity in the microbes decomposing the flies. Our work bridges a knowledge gap about how different substrates affect microbial decomposition, contributing to the broader understanding of ecosystem function in a nutrient cycling context.
Keywords: Sarracenia; bacterial function; decomposition; hydrolytic enzyme activity; microbial communities; pitcher plant.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


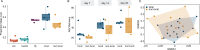
Similar articles
-
Selective Bacterial Community Enrichment between the Pitcher Plants Sarracenia minor and Sarracenia flava.Microbiol Spectr. 2021 Dec 22;9(3):e0069621. doi: 10.1128/Spectrum.00696-21. Epub 2021 Nov 24. Microbiol Spectr. 2021. PMID: 34817222 Free PMC article.
-
Regulation of Hydrolytic Enzyme Activity in Aquatic Microbial Communities Hosted by Carnivorous Pitcher Plants.Microb Ecol. 2018 Nov;76(4):885-898. doi: 10.1007/s00248-018-1187-z. Epub 2018 Apr 20. Microb Ecol. 2018. PMID: 29679120
-
An inquiline mosquito modulates microbial diversity and function in an aquatic microecosystem.Mol Ecol. 2024 Apr;33(7):e17314. doi: 10.1111/mec.17314. Epub 2024 Mar 5. Mol Ecol. 2024. PMID: 38441172
-
Traps of carnivorous pitcher plants as a habitat: composition of the fluid, biodiversity and mutualistic activities.Ann Bot. 2011 Feb;107(2):181-94. doi: 10.1093/aob/mcq238. Epub 2010 Dec 15. Ann Bot. 2011. PMID: 21159782 Free PMC article. Review.
-
Drivers and consequences of microbial community coalescence.ISME J. 2024 Jan 8;18(1):wrae179. doi: 10.1093/ismejo/wrae179. ISME J. 2024. PMID: 39288091 Free PMC article. Review.
References
-
- Nemergut DR, Cleveland CC, Wieder WR, Washenberger CL, Townsend AR. 2010. Plot-scale manipulations of organic matter inputs to soils correlate with shifts in microbial community composition in a lowland tropical rain forest. Soil Biol Biochem 42:2153–2160. doi:10.1016/j.soilbio.2010.08.011 - DOI
-
- Burns RG, Dick RP. 2002. Enzymes in the environment: activity, ecology, and applications. CRC Press.
-
- Bhatnagar JM, Peay KG, Treseder KK. 2018. Litter chemistry influences decomposition through activity of specific microbial functional guilds. Ecol Monogr 88:429–444. doi:10.1002/ecm.1303 - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

