Testicular differentiation in 46,XX DSD: an overview of genetic causes
- PMID: 38721146
- PMCID: PMC11076692
- DOI: 10.3389/fendo.2024.1385901
Testicular differentiation in 46,XX DSD: an overview of genetic causes
Abstract
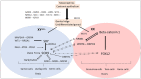
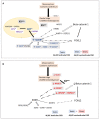
In mammals, the development of male or female gonads from fetal bipotential gonads depends on intricate genetic networks. Changes in dosage or temporal expression of sex-determining genes can lead to differences of gonadal development. Two rare conditions are associated with disruptions in ovarian determination, including 46,XX testicular differences in sex development (DSD), in which the 46,XX gonads differentiate into testes, and 46,XX ovotesticular DSD, characterized by the coexistence of ovarian and testicular tissue in the same individual. Several mechanisms have been identified that may contribute to the development of testicular tissue in XX gonads. This includes translocation of SRY to the X chromosome or an autosome. In the absence of SRY, other genes associated with testis development may be overexpressed or there may be a reduction in the activity of pro-ovarian/antitesticular factors. However, it is important to note that a significant number of patients with these DSD conditions have not yet recognized a genetic diagnosis. This finding suggests that there are additional genetic pathways or epigenetic mechanisms that have yet to be identified. The text will provide an overview of the current understanding of the genetic factors contributing to 46,XX DSD, specifically focusing on testicular and ovotesticular DSD conditions. It will summarize the existing knowledge regarding the genetic causes of these differences. Furthermore, it will explore the potential involvement of other factors, such as epigenetic mechanisms, in developing these conditions.
Keywords: 46; XX ovotesticular DSD; XX testicular DSD; differences of sex development (DSD); gonadal development; ovary.
Copyright © 2024 Ferrari, Silva, Nishi, Batista, Mendonca and Domenice.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Genetics, X-Linked Inheritance.2023 May 1. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2023 May 1. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 32491315 Free Books & Documents.
-
Immediate Hypersensitivity Reactions (Archived).2023 May 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2023 May 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30020687 Free Books & Documents.
-
Gadolinium Magnetic Resonance Imaging.2023 Jul 3. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2023 Jul 3. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 29494094 Free Books & Documents.
-
Depressing time: Waiting, melancholia, and the psychoanalytic practice of care.In: Kirtsoglou E, Simpson B, editors. The Time of Anthropology: Studies of Contemporary Chronopolitics. Abingdon: Routledge; 2020. Chapter 5. In: Kirtsoglou E, Simpson B, editors. The Time of Anthropology: Studies of Contemporary Chronopolitics. Abingdon: Routledge; 2020. Chapter 5. PMID: 36137063 Free Books & Documents. Review.
-
Ambiguous Genitalia in the Newborn.2023 Nov 13. In: Feingold KR, Anawalt B, Blackman MR, Boyce A, Chrousos G, Corpas E, de Herder WW, Dhatariya K, Dungan K, Hofland J, Kalra S, Kaltsas G, Kapoor N, Koch C, Kopp P, Korbonits M, Kovacs CS, Kuohung W, Laferrère B, Levy M, McGee EA, McLachlan R, Muzumdar R, Purnell J, Sahay R, Shah AS, Singer F, Sperling MA, Stratakis CA, Trence DL, Wilson DP, editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000–. 2023 Nov 13. In: Feingold KR, Anawalt B, Blackman MR, Boyce A, Chrousos G, Corpas E, de Herder WW, Dhatariya K, Dungan K, Hofland J, Kalra S, Kaltsas G, Kapoor N, Koch C, Kopp P, Korbonits M, Kovacs CS, Kuohung W, Laferrère B, Levy M, McGee EA, McLachlan R, Muzumdar R, Purnell J, Sahay R, Shah AS, Singer F, Sperling MA, Stratakis CA, Trence DL, Wilson DP, editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000–. PMID: 25905391 Free Books & Documents. Review.
Cited by
-
A Case Report on 46,XX Male Difference of Sex Development.Cureus. 2024 Sep 5;16(9):e68741. doi: 10.7759/cureus.68741. eCollection 2024 Sep. Cureus. 2024. PMID: 39371808 Free PMC article.
References
-
- Osorio Acosta VA, Alonso Domínguez FJ. [True hermaphroditism]. Arch Esp Urol. (2004) 57:856–60. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

