A Phase I Study of KIN-3248, an Irreversible Small-molecule Pan-FGFR Inhibitor, in Patients with Advanced FGFR2/3-driven Solid Tumors
- PMID: 38602417
- PMCID: PMC11060137
- DOI: 10.1158/2767-9764.CRC-24-0137
A Phase I Study of KIN-3248, an Irreversible Small-molecule Pan-FGFR Inhibitor, in Patients with Advanced FGFR2/3-driven Solid Tumors
Abstract
Purpose: Despite efficacy of approved FGFR inhibitors, emergence of polyclonal secondary mutations in the FGFR kinase domain leads to acquired resistance. KIN-3248 is a selective, irreversible, orally bioavailable, small-molecule inhibitor of FGFR1-4 that blocks both primary oncogenic and secondary kinase domain resistance FGFR alterations.
Experimental design: A first-in-human, phase I study of KIN-3248 was conducted in patients with advanced solid tumors harboring FGFR2 and/or FGFR3 gene alterations (NCT05242822). The primary objective was determination of MTD/recommended phase II dose (RP2D). Secondary and exploratory objectives included antitumor activity, pharmacokinetics, pharmacodynamics, and molecular response by circulating tumor DNA (ctDNA) clearance.
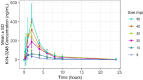
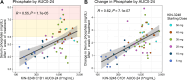
Results: Fifty-four patients received doses ranging from 5 to 50 mg orally daily across six cohorts. Intrahepatic cholangiocarcinoma (48.1%), gastric (9.3%), and urothelial (7.4%) were the most common tumors. Tumors harbored FGFR2 (68.5%) or FGFR3 (31.5%) alterations-23 (42.6%) received prior FGFR inhibitors. One dose-limiting toxicity (hypersensitivity) occurred in cohort 1 (5 mg). Treatment-related, adverse events included hyperphosphatemia, diarrhea, and stomatitis. The MTD/RP2D was not established. Exposure was dose proportional and concordant with hyperphosphatemia. Five partial responses were observed; 4 in FGFR inhibitor naïve and 1 in FGFR pretreated patients. Pretreatment ctDNA profiling confirmed FGFR2/3 alterations in 63.3% of cases and clearance at cycle 2 associated with radiographic response.
Conclusion: The trial was terminated early for commercial considerations; therefore, RP2D was not established. Preliminary clinical data suggest that KIN-3248 is a safe, oral FGFR1-4 inhibitor with favorable pharmacokinetic parameters, though further dose escalation was required to nominate the MTD/RP2D.
Significance: KIN-3248 was a rationally designed, next generation selective FGFR inhibitor, that was effective in interfering with both FGFR wild-type and mutant signaling. Clinical data indicate that KIN-3248 is safe with a signal of antitumor activity. Translational science support the mechanism of action in that serum phosphate was proportional with exposure, paired biopsies suggested phospho-ERK inhibition (a downstream target of FGFR2/3), and ctDNA clearance may act as a RECIST response surrogate.
© 2024 The Authors; Published by the American Association for Cancer Research.
Figures

Similar articles
-
The Irreversible FGFR Inhibitor KIN-3248 Overcomes FGFR2 Kinase Domain Mutations.Clin Cancer Res. 2024 May 15;30(10):2181-2192. doi: 10.1158/1078-0432.CCR-23-3588. Clin Cancer Res. 2024. PMID: 38437671 Free PMC article.
-
Can a Liquid Biopsy Detect Circulating Tumor DNA With Low-passage Whole-genome Sequencing in Patients With a Sarcoma? A Pilot Evaluation.Clin Orthop Relat Res. 2025 Jan 1;483(1):39-48. doi: 10.1097/CORR.0000000000003161. Epub 2024 Jun 21. Clin Orthop Relat Res. 2025. PMID: 38905450
-
Tasurgratinib in patients with cholangiocarcinoma or gastric cancer: Expansion part of the first-in-human phase I study.Cancer Sci. 2025 Jan;116(1):192-203. doi: 10.1111/cas.16354. Epub 2024 Oct 27. Cancer Sci. 2025. PMID: 39462221 Free PMC article. Clinical Trial.
-
Oral budesonide for induction of remission in ulcerative colitis.Cochrane Database Syst Rev. 2015 Oct 26;2015(10):CD007698. doi: 10.1002/14651858.CD007698.pub3. Cochrane Database Syst Rev. 2015. PMID: 26497719 Free PMC article. Review.
-
Impact of residual disease as a prognostic factor for survival in women with advanced epithelial ovarian cancer after primary surgery.Cochrane Database Syst Rev. 2022 Sep 26;9(9):CD015048. doi: 10.1002/14651858.CD015048.pub2. Cochrane Database Syst Rev. 2022. PMID: 36161421 Free PMC article. Review.
Cited by
-
FGFR2-fusions define a clinically actionable molecular subset of pancreatic cancer.NPJ Precis Oncol. 2024 Sep 17;8(1):207. doi: 10.1038/s41698-024-00683-x. NPJ Precis Oncol. 2024. PMID: 39289482 Free PMC article.
References
-
- Babina IS, Turner NC. Advances and challenges in targeting FGFR signalling in cancer. Nat Rev Cancer 2017;17:318–32. - PubMed
-
- Touat M, Ileana E, Postel-Vinay S, Andre F, Soria JC. Targeting FGFR signaling in cancer. Clin Cancer Res 2015;21:2684–94. - PubMed
-
- Loriot Y, Necchi A, Park SH, Garcia-Donas J, Huddart R, Burgess E, et al. . Erdafitinib in locally advanced or metastatic urothelial carcinoma. N Engl J Med 2019;381:338–48. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

