Combination of AS101 and Mefloquine Inhibits Carbapenem-Resistant Pseudomonas aeruginosa in vitro and in vivo
- PMID: 38023412
- PMCID: PMC10664714
- DOI: 10.2147/IDR.S427232
Combination of AS101 and Mefloquine Inhibits Carbapenem-Resistant Pseudomonas aeruginosa in vitro and in vivo
Abstract
Background: In recent years, carbapenem-resistant Pseudomonas aeruginosa (CRPA) has spread around the world, leading to a high mortality and close attention of medical community. In this study, we aim to find a new strategy of treatment for CRPA infections.
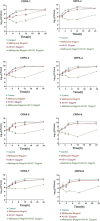
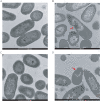
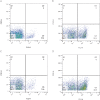
Methods: Eight strains of CRPA were collected, and PCR detected the multi-locus sequence typing (MLST). The antimicrobial susceptibility test was conducted using the VITEK@2 compact system. The minimum inhibitory concentration (MIC) for AS101 and mefloquine was determined using the broth dilution method. Antibacterial activity was tested in vitro and in vivo through the chessboard assay, time killing assay, and a mouse model. The mechanism of AS101 combined with mefloquine against CRPA was assessed through the biofilm formation inhibition assay, electron microscopy, and detection of reactive oxygen species (ROS).
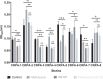
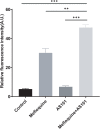
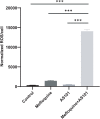
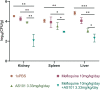
Results: The results demonstrated that all tested CRPA strains exhibited multidrug resistance. Moreover, our investigation revealed a substantial synergistic antibacterial effect of AS101-mefloquine in vitro. The assay for inhibiting biofilm formation indicated that AS101-mefloquine effectively suppressed the biofilm formation of CRPA-5 and CRPA-6. Furthermore, AS101-mefloquine were observed to disrupt the bacterial cell wall and enhance the permeability of the cell membrane. This effect was achieved by stimulating the production of ROS, which in turn hindered the growth of CRPA-3. To evaluate the therapeutic potential, a murine model of CRPA-3 peritoneal infection was established. Notably, AS101-mefloquine administration resulted in a significant reduction in bacterial load within the liver, kidney, and spleen of mice after 72 hours of treatment.
Conclusion: The present study showed that the combination of AS101 and mefloquine yielded a notable synergistic bacteriostatic effect both in vitro and in vivo, suggesting a potential clinical application of this combination in the treatment of CRPA.
Keywords: AS101; Pseudomonas aeruginosa; biofilm; carbapenem resistance; mefloquine; synergy.
© 2023 Li et al.
Conflict of interest statement
The authors have no competing interests to declare for this work.
Figures

Similar articles
-
In Vitro and In Vivo Activity of AS101 against Carbapenem-Resistant Acinetobacter baumannii.Pharmaceuticals (Basel). 2021 Aug 21;14(8):823. doi: 10.3390/ph14080823. Pharmaceuticals (Basel). 2021. PMID: 34451920 Free PMC article.
-
Combined With Mefloquine, Resurrect Colistin Active in Colistin-Resistant Pseudomonas aeruginosa in vitro and in vivo.Front Microbiol. 2021 Nov 26;12:790220. doi: 10.3389/fmicb.2021.790220. eCollection 2021. Front Microbiol. 2021. PMID: 34899672 Free PMC article.
-
In vitro and in vivo antibiofilm activity of the synthetic antimicrobial peptide WLBU2 against multiple drug resistant Pseudomonas aeruginosa strains.BMC Microbiol. 2023 May 15;23(1):131. doi: 10.1186/s12866-023-02886-x. BMC Microbiol. 2023. PMID: 37183241 Free PMC article.
-
Antibacterial and antibiofilm activities of fosfomycin combined with rifampin against carbapenem-resistant Pseudomonas aeruginosa.Lett Appl Microbiol. 2022 Dec;75(6):1559-1568. doi: 10.1111/lam.13822. Epub 2022 Sep 23. Lett Appl Microbiol. 2022. PMID: 36036376
-
Molecular epidemiology of carbapenem-resistant Pseudomonas aeruginosa in an endemic area: comparison with global data.Eur J Clin Microbiol Infect Dis. 2018 Jul;37(7):1211-1220. doi: 10.1007/s10096-018-3244-4. Epub 2018 Apr 11. Eur J Clin Microbiol Infect Dis. 2018. PMID: 29644540 Review.
References
-
- Mirzaei B, Bazgir ZN, Goli HR, et al. Prevalence of multi-drug resistant (MDR) and extensively drug-resistant (XDR) phenotypes of Pseudomonas aeruginosa and Acinetobacter baumannii isolated in clinical samples from northeast of Iran. BMC Res Notes. 2020;13(1):380. doi:10.1186/s13104-020-05224-w - DOI - PMC - PubMed
-
- CDC. Antibiotic resistance threats in the United States; 2019. https://www.cdc.gov/drugresistance/pdf/threats-report/2019-ar-threats-re.... Accessed November 01, 2023.
Grants and funding
LinkOut - more resources
Full Text Sources

