Alterations in Antibiotic Susceptibility of Staphylococcus aureus and Klebsiella pneumoniae in Dual Species Biofilms
- PMID: 37239822
- PMCID: PMC10217825
- DOI: 10.3390/ijms24108475
Alterations in Antibiotic Susceptibility of Staphylococcus aureus and Klebsiella pneumoniae in Dual Species Biofilms
Abstract
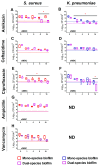
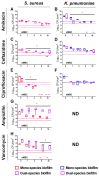
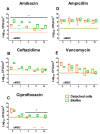
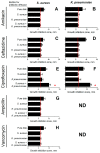
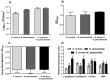
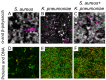
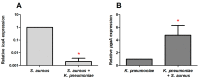
In the last decades, it has been shown that biofilm-associated infections in most cases are caused by rather two or even more pathogens than by single microorganisms. Due to intermicrobial interactions in mixed communities, bacteria change their gene expression profile, in turn leading to alterations in the biofilm structure and properties, as well as susceptibility to antimicrobials. Here, we report the alterations of antimicrobials efficiency in mixed biofilms of Staphylococcus aureus-Klebsiella pneumoniae in comparison with mono-species biofilms of each counterpart and discuss possible mechanisms of these alterations. In cell clumps detached from dual-species biofilms, S. aureus became insensitive to vancomycin, ampicillin, and ceftazidime compared to solely S. aureus cell clumps. In turn, the increased efficiency of amikacin and ciprofloxacin against both bacteria could be observed, compared to mono-species biofilms of each counterpart. Scanning electron microscopy and confocal microscopy indicate the porous structure of the dual-species biofilm, and differential fluorescent staining revealed an increased number of polysaccharides in the matrix, in turn leading to more loose structure and thus apparently providing increased permeability of the dual-species biofilm to antimicrobials. The qRT-PCR showed that ica operon in S. aureus became repressed in mixed communities, and polysaccharides are produced mainly by K. pneumoniae. While the molecular trigger of these changes remains undiscovered, detailed knowledge of the alterations in antibiotic susceptibility to given drugs opens doors for treatment correction options for S. aureus-K. pneumoniae biofilm-associated infections.
Keywords: biochemical composition; dual-species biofilms; extracellular matrix; gene expression; sensitivity to antimicrobials.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Effect of Antimicrobial and Physical Treatments on Growth of Multispecies Staphylococcal Biofilms.Appl Environ Microbiol. 2017 May 31;83(12):e03483-16. doi: 10.1128/AEM.03483-16. Print 2017 Jun 15. Appl Environ Microbiol. 2017. PMID: 28411222 Free PMC article.
-
Interspecies relationships between nosocomial pathogens associated to preterm infants and lactic acid bacteria in dual-species biofilms.Front Cell Infect Microbiol. 2022 Oct 17;12:1038253. doi: 10.3389/fcimb.2022.1038253. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 36325465 Free PMC article.
-
Matrix Polysaccharides and SiaD Diguanylate Cyclase Alter Community Structure and Competitiveness of Pseudomonas aeruginosa during Dual-Species Biofilm Development with Staphylococcus aureus.mBio. 2018 Nov 6;9(6):e00585-18. doi: 10.1128/mBio.00585-18. mBio. 2018. PMID: 30401769 Free PMC article.
-
Vision for medicine: Staphylococcus aureus biofilm war and unlocking key's for anti-biofilm drug development.Microb Pathog. 2018 Oct;123:339-347. doi: 10.1016/j.micpath.2018.07.002. Epub 2018 Jul 3. Microb Pathog. 2018. PMID: 30057355 Review.
-
Klebsiella pneumoniae Biofilms and Their Role in Disease Pathogenesis.Front Cell Infect Microbiol. 2022 May 11;12:877995. doi: 10.3389/fcimb.2022.877995. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 35646720 Free PMC article. Review.
Cited by
-
Microbial Biofilms: Features of Formation and Potential for Use in Bioelectrochemical Devices.Biosensors (Basel). 2024 Jun 8;14(6):302. doi: 10.3390/bios14060302. Biosensors (Basel). 2024. PMID: 38920606 Free PMC article. Review.
-
The Antimicrobial Potential of the Hop (Humulus lupulus L.) Extract against Staphylococcus aureus and Oral Streptococci.Pharmaceuticals (Basel). 2024 Jan 27;17(2):162. doi: 10.3390/ph17020162. Pharmaceuticals (Basel). 2024. PMID: 38399377 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

