Safety and Immunogenicity of an Investigational Respiratory Syncytial Virus Vaccine (RSVPreF3) in Mothers and Their Infants: A Phase 2 Randomized Trial
- PMID: 36722147
- PMCID: PMC10420396
- DOI: 10.1093/infdis/jiad024
Safety and Immunogenicity of an Investigational Respiratory Syncytial Virus Vaccine (RSVPreF3) in Mothers and Their Infants: A Phase 2 Randomized Trial
Abstract
Background: In a phase 1/2 study, a maternal respiratory syncytial virus vaccine candidate (RSVPreF3) demonstrated an acceptable safety profile and efficiently increased RSV-specific humoral immune responses in non-pregnant women.
Methods: In this phase 2 observer-blind, placebo-controlled, randomized clinical trial (NCT04126213), the safety of RSVPreF3 (60 or 120 µg), administered during late second or third trimester, was evaluated in 213 18- to 40-year-old healthy pregnant women through 6 months postdelivery and their offspring through infancy; immunogenicity was evaluated through day 43 postdelivery and day 181 postbirth, respectively.
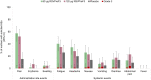
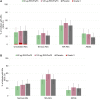
Results: RSVPreF3 was well tolerated. No pregnancy-related or neonatal adverse events of special interest were considered vaccine/placebo related. In the 60 and 120 µg RSVPreF3 groups: (1) neutralizing antibody (nAb) titers in mothers increased 12.7- and 14.9-fold against RSV-A and 10.6- and 13.2-fold against RSV-B, respectively, 1 month postvaccination and remained 8.9-10.0-fold over prevaccination at day 43 postdelivery; (2) nAb titers were consistently higher compared to placebo recipients; (3) placental transfer ratios for anti-RSVPreF3 antibodies at birth were 1.62 and 1.90, respectively, and (4) nAb levels in infants were highest at birth and declined through day 181 postbirth.
Conclusions: RSVPreF3 maternal vaccination had an acceptable safety risk profile and induced robust RSV-specific immune responses with successful antibody transfer to their newborns.
Clinical trials registration: NCT04126213.
Keywords: RSV; fetal care; humoral immunity; maternal immunization; neonatal care; passive immunization; placental transfer; pregnancy; preterm birth.
Plain language summary
What is the context?: Infants, especially those less than 6 months of age, are at increased risk of lung infection caused by respiratory syncytial virus (RSV). However, this risk could be reduced with maternal vaccination against RSV during pregnancy. A previous clinical trial found that a vaccine candidate (named RSVPreF3) was well tolerated when given to non-pregnant women.
What is new?: In pregnant women, RSVPreF3 was also well tolerated. Occurrence of unsolicited adverse events was similar between vaccine and placebo recipients. None of the serious adverse events or events of interest for pregnant women or newborns were considered related to the study intervention. One month after vaccination, mothers who received RSVPreF3 had 11–15 times higher levels of antibodies against RSV than before vaccination. These antibody levels remained similar until 43 days after delivery. In the infants born to mothers vaccinated during pregnancy with RSVPreF3, antibody levels were highest at birth, when levels were higher than in their mothers, and declined through day 181 postbirth.
What is the impact?: RSVPreF3 had an acceptable safety risk profile in pregnant women and their babies. This vaccine induced potent immune responses against RSV, with maternal antibodies transferred to infants of the vaccinated mothers.
© The Author(s) 2023. Published by Oxford University Press on behalf of Infectious Diseases Society of America.
Conflict of interest statement
Potential conflicts of interest. A. N. T., H. Q., I. D., M. H., M. d. H., O. H., P. L., M. M. C., and Z. B. are or were employees of the GSK group of companies at the time of the study. A. N. T., I. D., M. d. H., M. M. C., and O. H. report holding shares of the GSK group of companies. P. L. reports personal fees from GSK. O. R. declares personal fees from CEVAXIN during the conduct of the study and outside the submitted work. A. K. declares institutional financial support from GSK for conducting this study; investigator-initiated grants from Valneva and Pfizer, the latter two outside this study; and declares no personal fees from any vaccine companies. M. G. S. declares personal fees and institutional financial support from GSK during the conduct of the study. J. M. L. declares holding the Canadian Institutes of Health Research-GSK Chair in Pediatric Vaccinology at Dalhousie University; institutional financial support from GSK during the conduct of the study; other compensation from Immunovaccine; and grants from Novavax, Janssen, and Regeneron, outside the submitted work; in all cases these are paid to Dalhousie University. J. B. declares institutional financial support from GSK, Pfizer, and MedImmune during the conduct of the study and outside the submitted work. S. A. M. has received institution grants and personal fees from BMGF and GSK; and institution grants from Pfizer, Novavax, AstraZeneca, and Minervax during the conduct of the study. R. G. declares personal fees from Optimal Clinical Trials during the conduct of the study. All other authors report no potential conflicts. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
Figures



Similar articles
-
Safety and immunogenicity of a respiratory syncytial virus prefusion F protein (RSVPreF3) candidate vaccine in older Japanese adults: A phase I, randomized, observer-blind clinical trial.Respir Investig. 2023 Mar;61(2):261-269. doi: 10.1016/j.resinv.2022.11.003. Epub 2023 Jan 12. Respir Investig. 2023. PMID: 36641341 Clinical Trial.
-
Safety and Immunogenicity of a Revaccination With a Respiratory Syncytial Virus Prefusion F Vaccine in Older Adults: A Phase 2b Study.J Infect Dis. 2024 Feb 14;229(2):355-366. doi: 10.1093/infdis/jiad321. J Infect Dis. 2024. PMID: 37699064 Free PMC article.
-
Three Dose Levels of a Maternal Respiratory Syncytial Virus Vaccine Candidate Are Well Tolerated and Immunogenic in a Randomized Trial in Nonpregnant Women.J Infect Dis. 2022 Jun 15;225(12):2067-2076. doi: 10.1093/infdis/jiab317. J Infect Dis. 2022. PMID: 34146100 Free PMC article. Clinical Trial.
-
Passive protection against respiratory syncytial virus disease in infants: the role of maternal antibody.Pediatr Infect Dis J. 1994 May;13(5):449-53. doi: 10.1097/00006454-199405000-00037. Pediatr Infect Dis J. 1994. PMID: 8072835 Review.
-
Profile of respiratory syncytial virus prefusogenic fusion protein nanoparticle vaccine.Expert Rev Vaccines. 2021 Apr;20(4):351-364. doi: 10.1080/14760584.2021.1903877. Epub 2021 May 2. Expert Rev Vaccines. 2021. PMID: 33733995 Review.
Cited by
-
Recent advances in the prevention of respiratory syncytial virus in pediatrics.Curr Opin Pediatr. 2024 Apr 1;36(2):182-189. doi: 10.1097/MOP.0000000000001336. Epub 2024 Jan 30. Curr Opin Pediatr. 2024. PMID: 38299987 Review.
-
Maternal Vaccination for the Prevention of Infantile RSV Disease: An Overview of the Authorized, In-Progress, and Rejected Vaccine Candidates.Vaccines (Basel). 2024 Aug 28;12(9):980. doi: 10.3390/vaccines12090980. Vaccines (Basel). 2024. PMID: 39340012 Free PMC article. Review.
-
Use of the Abrysvo Vaccine in Pregnancy to Prevent Respiratory Syncytial Virus in Infants: A Review.Cureus. 2024 Aug 31;16(8):e68349. doi: 10.7759/cureus.68349. eCollection 2024 Aug. Cureus. 2024. PMID: 39355078 Free PMC article. Review.
-
Respiratory syncytial virus vaccination during pregnancy for improving infant outcomes.Cochrane Database Syst Rev. 2024 May 2;5(5):CD015134. doi: 10.1002/14651858.CD015134.pub2. Cochrane Database Syst Rev. 2024. PMID: 38695784 Review.
-
Characterisation of RSV infections in children without chronic diseases aged 0-36 months during the post-COVID-19 winter season 2022/2023.Front Pediatr. 2024 Feb 6;12:1342399. doi: 10.3389/fped.2024.1342399. eCollection 2024. Front Pediatr. 2024. PMID: 38379907 Free PMC article.
References
-
- Jorquera PA, Tripp RA. Respiratory syncytial virus: prospects for new and emerging therapeutics. Expert Rev Respir Med 2017; 11:609–15. - PubMed
-
- American Academy of Pediatrics . Updated guidance for palivizumab prophylaxis among infants and young children at increased risk of hospitalization for respiratory syncytial virus infection. Pediatrics 2014; 134:e620–38. - PubMed
-
- American Academy of Pediatrics . Diagnosis and management of bronchiolitis. Pediatrics 2006; 118:1774–93. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

