Deucravacitinib, a Tyrosine Kinase 2 Inhibitor, in Systemic Lupus Erythematosus: A Phase II, Randomized, Double-Blind, Placebo-Controlled Trial
- PMID: 36369798
- PMCID: PMC10100399
- DOI: 10.1002/art.42391
Deucravacitinib, a Tyrosine Kinase 2 Inhibitor, in Systemic Lupus Erythematosus: A Phase II, Randomized, Double-Blind, Placebo-Controlled Trial
Abstract
Objective: To assess the efficacy and safety of deucravacitinib, an oral, selective, allosteric inhibitor of TYK2, in a phase II trial in adult patients with active systemic lupus erythematosus (SLE).
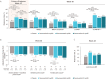
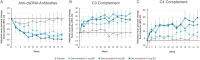
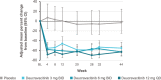
Methods: Adults with active SLE were enrolled from 162 sites in 17 countries. Patients (n = 363) were randomized 1:1:1:1 to receive deucravacitinib 3 mg twice daily, 6 mg twice daily, 12 mg once daily, or placebo. The primary end point was SLE Responder Index 4 (SRI-4) response at week 32. Secondary outcomes assessed at week 48 included SRI-4, British Isles Lupus Assessment Group-based Composite Lupus Assessment (BICLA) response, Cutaneous Lupus Erythematosus Disease Area and Severity Index 50 (CLASI-50), Lupus Low Disease Activity State (LLDAS), and improvements in active (swollen plus tender), swollen, and tender joint counts.
Results: At week 32, the percentage of patients achieving SRI-4 response was 34% with placebo compared to 58% with deucravacitinib 3 mg twice daily (odds ratio [OR] 2.8 [95% confidence interval (95% CI) 1.5, 5.1]; P < 0.001 versus placebo), 50% with 6 mg twice daily (OR 1.9 [95% CI 1.0, 3.4]; P = 0.02 versus placebo), and 45% with 12 mg once daily (OR 1.6 [95% CI 0.8, 2.9]; nominal P = 0.08 versus placebo). Response rates were higher with deucravacitinib treatment for BICLA, CLASI-50, LLDAS, and joint counts compared to placebo. Rates of adverse events were similar across groups, except higher rates of infections and cutaneous events, including rash and acne, with deucravacitinib treatment. Rates of serious adverse events were comparable, with no deaths, opportunistic infections, tuberculosis infections, major adverse cardiovascular events, or thrombotic events reported.
Conclusion: Deucravacitinib treatment elicited higher response rates for SRI-4 and other end points compared with placebo, with an acceptable safety profile, in adult patients with active SLE.
Trial registration: ClinicalTrials.gov NCT03252587.
© 2022 The Authors. Arthritis & Rheumatology published by Wiley Periodicals LLC on behalf of American College of Rheumatology.
Figures

Similar articles
-
Interferon Inhibition for Lupus with Anifrolumab: Critical Appraisal of the Evidence Leading to FDA Approval.ACR Open Rheumatol. 2022 Jun;4(6):486-491. doi: 10.1002/acr2.11414. Epub 2022 Feb 14. ACR Open Rheumatol. 2022. PMID: 35157371 Free PMC article.
-
Type I interferon inhibitor anifrolumab in active systemic lupus erythematosus (TULIP-1): a randomised, controlled, phase 3 trial.Lancet Rheumatol. 2019 Dec;1(4):e208-e219. doi: 10.1016/S2665-9913(19)30076-1. Epub 2019 Nov 11. Lancet Rheumatol. 2019. PMID: 38229377
-
Efficacy, Safety, and Pharmacodynamic Effects of the Bruton's Tyrosine Kinase Inhibitor Fenebrutinib (GDC-0853) in Systemic Lupus Erythematosus: Results of a Phase II, Randomized, Double-Blind, Placebo-Controlled Trial.Arthritis Rheumatol. 2021 Oct;73(10):1835-1846. doi: 10.1002/art.41811. Epub 2021 Aug 24. Arthritis Rheumatol. 2021. PMID: 34042314 Clinical Trial.
-
Efficacy and safety of Janus kinase inhibitors in systemic and cutaneous lupus erythematosus: A systematic review and meta-analysis.Autoimmun Rev. 2023 Dec;22(12):103440. doi: 10.1016/j.autrev.2023.103440. Epub 2023 Sep 9. Autoimmun Rev. 2023. PMID: 37678618 Review.
-
Deucravacitinib: The First FDA-Approved Oral TYK2 Inhibitor for Moderate to Severe Plaque Psoriasis.Ann Pharmacother. 2024 Apr;58(4):416-427. doi: 10.1177/10600280231153863. Epub 2023 Jun 21. Ann Pharmacother. 2024. PMID: 37341177 Review.
Cited by
-
B Cell Tolerance and Targeted Therapies in SLE.J Clin Med. 2023 Sep 28;12(19):6268. doi: 10.3390/jcm12196268. J Clin Med. 2023. PMID: 37834911 Free PMC article. Review.
-
Deucravacitinib, a tyrosine kinase 2 pseudokinase inhibitor, protects human EndoC-βH1 β-cells against proinflammatory insults.Front Immunol. 2023 Oct 3;14:1263926. doi: 10.3389/fimmu.2023.1263926. eCollection 2023. Front Immunol. 2023. PMID: 37854597 Free PMC article.
-
JAK Inhibition as a Potential Treatment Target in Systemic Lupus Erythematosus.Mediterr J Rheumatol. 2024 Mar 30;35(Suppl 1):37-44. doi: 10.31138/mjr.231123.jia. eCollection 2024 Mar. Mediterr J Rheumatol. 2024. PMID: 38756931 Free PMC article. Review.
-
Systemic lupus erythematosus: pathogenesis and targeted therapy.Mol Biomed. 2024 Oct 30;5(1):54. doi: 10.1186/s43556-024-00217-8. Mol Biomed. 2024. PMID: 39472388 Free PMC article. Review.
-
Targeted Therapy for SLE-What Works, What Doesn't, What's Next.J Clin Med. 2023 Apr 29;12(9):3198. doi: 10.3390/jcm12093198. J Clin Med. 2023. PMID: 37176637 Free PMC article. Review.
References
-
- Kaul A, Gordon C, Crow MK, et al. Systemic lupus erythematosus [review]. Nat Rev Dis Primers 2016;2:16039. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

