Margetuximab Versus Trastuzumab in Patients With Previously Treated HER2-Positive Advanced Breast Cancer (SOPHIA): Final Overall Survival Results From a Randomized Phase 3 Trial
- PMID: 36332179
- PMCID: PMC9839304
- DOI: 10.1200/JCO.21.02937
Margetuximab Versus Trastuzumab in Patients With Previously Treated HER2-Positive Advanced Breast Cancer (SOPHIA): Final Overall Survival Results From a Randomized Phase 3 Trial
Abstract
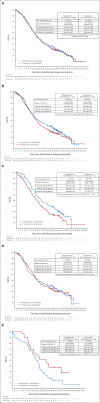
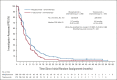
Clinical trials frequently include multiple end points that mature at different times. The initial report, typically based on the primary end point, may be published when key planned co-primary or secondary analyses are not yet available. Clinical Trial Updates provide an opportunity to disseminate additional results from studies, published in JCO or elsewhere, for which the primary end point has already been reported.Final overall survival (OS) in SOPHIA (ClinicalTrials.gov identifier: NCT02492711), a study of margetuximab versus trastuzumab, both with chemotherapy, in patients with previously treated human epidermal growth factor receptor 2-positive advanced breast cancer, is reported with updated safety. Overall, 536 patients in the intention-to-treat population were randomly assigned to margetuximab (15 mg/kg intravenously once every 3 weeks; n = 266) plus chemotherapy or trastuzumab (6 mg/kg intravenously once every 3 weeks after a loading dose of 8 mg/kg; n = 270) plus chemotherapy. Primary end points were progression-free survival, previously reported, and OS. Final OS analysis was triggered by 385 prespecified events. The median OS was 21.6 months (95% CI, 18.89 to 25.07) with margetuximab versus 21.9 months (95% CI, 18.69 to 24.18) with trastuzumab (hazard ratio [HR], 0.95; 95% CI, 0.77 to 1.17; P = .620). Preplanned, exploratory analysis of CD16A genotyping suggested a possible improvement in OS for margetuximab in CD16A-158FF patients versus trastuzumab (median OS, 23.6 v 19.2 months; HR, 0.72; 95% CI, 0.52 to 1.00) and a possible improvement in OS for trastuzumab in CD16A-158VV patients versus margetuximab (median OS, 31.1 v 22.0 months; HR, 1.77; 95% CI, 1.01 to 3.12). Margetuximab safety was comparable with trastuzumab. Final overall OS analysis did not demonstrate margetuximab advantage over trastuzumab. Margetuximab studies in patients with human epidermal growth factor receptor 2-positive breast cancer with different CD16A allelic variants are warranted.
Conflict of interest statement
No other potential conflicts of interest were reported.
Figures

Similar articles
-
Efficacy of Margetuximab vs Trastuzumab in Patients With Pretreated ERBB2-Positive Advanced Breast Cancer: A Phase 3 Randomized Clinical Trial.JAMA Oncol. 2021 Apr 1;7(4):573-584. doi: 10.1001/jamaoncol.2020.7932. JAMA Oncol. 2021. PMID: 33480963 Free PMC article. Clinical Trial.
-
Adjuvant Pertuzumab and Trastuzumab in Early Human Epidermal Growth Factor Receptor 2-Positive Breast Cancer in the APHINITY Trial: Third Interim Overall Survival Analysis With Efficacy Update.J Clin Oncol. 2024 Nov;42(31):3643-3651. doi: 10.1200/JCO.23.02505. Epub 2024 Sep 11. J Clin Oncol. 2024. PMID: 39259927 Clinical Trial.
-
Margetuximab Versus Trastuzumab in Patients With Advanced Breast Cancer: A Cost-effectiveness Analysis.Clin Breast Cancer. 2022 Jul;22(5):e629-e635. doi: 10.1016/j.clbc.2022.03.002. Epub 2022 Mar 17. Clin Breast Cancer. 2022. PMID: 35418312
-
Trastuzumab-containing regimens for metastatic breast cancer.Cochrane Database Syst Rev. 2014 Jun 12;2014(6):CD006242. doi: 10.1002/14651858.CD006242.pub2. Cochrane Database Syst Rev. 2014. PMID: 24919460 Free PMC article. Review.
-
Margetuximab: First Approval.Drugs. 2021 Apr;81(5):599-604. doi: 10.1007/s40265-021-01485-2. Epub 2021 Mar 24. Drugs. 2021. PMID: 33761116 Review.
Cited by
-
A Review of Margetuximab-Based Therapies in Patients with HER2-Positive Metastatic Breast Cancer.Cancers (Basel). 2022 Dec 21;15(1):38. doi: 10.3390/cancers15010038. Cancers (Basel). 2022. PMID: 36612034 Free PMC article. Review.
-
Update on current and new potential immunotherapies in breast cancer, from bench to bedside.Front Immunol. 2024 Feb 15;15:1287824. doi: 10.3389/fimmu.2024.1287824. eCollection 2024. Front Immunol. 2024. PMID: 38433837 Free PMC article. Review.
-
Targeting CD276 for T cell-based immunotherapy of breast cancer.J Transl Med. 2024 Oct 4;22(1):902. doi: 10.1186/s12967-024-05689-4. J Transl Med. 2024. PMID: 39367484 Free PMC article.
-
Update Breast Cancer 2022 Part 5 - Early Stage Breast Cancer.Geburtshilfe Frauenheilkd. 2023 Mar 9;83(3):289-298. doi: 10.1055/a-2018-9053. eCollection 2023 Mar. Geburtshilfe Frauenheilkd. 2023. PMID: 36908285 Free PMC article.
-
Cardiac Safety and Efficacy of SB3 Trastuzumab Biosimilar for ERBB2-Positive Early Breast Cancer: Secondary Analysis of a Randomized Clinical Trial.JAMA Netw Open. 2023 Apr 3;6(4):e235822. doi: 10.1001/jamanetworkopen.2023.5822. JAMA Netw Open. 2023. PMID: 37022687 Free PMC article. Clinical Trial.
References
-
- Stavenhagen JB, Gorlatov S, Tuaillon N, et al. : Fc optimization of therapeutic antibodies enhances their ability to kill tumor cells in vitro and controls tumor expansion in vivo via low-affinity activating Fcgamma receptors. Cancer Res 67:8882-8890, 2007 - PubMed
-
- Liu L, Yang Y, Burns R, et al. : Margetuximab mediates greater Fc-dependent anti-tumor activities than trastuzumab or pertuzumab in vitro. Cancer Res 79:1538, 2019 (abstr 1538)
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

