Hydroxyurea (hydroxycarbamide) for sickle cell disease
- PMID: 36047926
- PMCID: PMC9435593
- DOI: 10.1002/14651858.CD002202.pub3
Hydroxyurea (hydroxycarbamide) for sickle cell disease
Abstract
Background: Sickle cell disease (SCD) is one of the most common inherited diseases worldwide. It is associated with lifelong morbidity and a reduced life expectancy. Hydroxyurea (hydroxycarbamide), an oral chemotherapeutic drug, ameliorates some of the clinical problems of SCD, in particular that of pain, by raising foetal haemoglobin (HbF). This is an update of a previously published Cochrane Review.
Objectives: The aims of this review are to determine through a review of randomised or quasi-randomised studies whether the use of hydroxyurea in people with SCD alters the pattern of acute events, including pain; prevents, delays or reverses organ dysfunction; alters mortality and quality of life; or is associated with adverse effects. In addition, we hoped to assess whether the response to hydroxyurea in SCD varies with the type of SCD, age of the individual, duration and dose of treatment, and healthcare setting.
Search methods: We searched the Cochrane Cystic Fibrosis and Genetic Disorders Haemoglobinopathies Register, comprising references identified from comprehensive electronic database searches and handsearches of relevant journals and abstract books of conference proceedings. We also searched online trial registries. The date of the most recent search was 17 February 2022.
Selection criteria: Randomised and quasi-randomised controlled trials (RCTs and quasi-RCTs), of one month or longer, comparing hydroxyurea with placebo or standard therapy in people with SCD.
Data collection and analysis: Authors independently assessed studies for inclusion, carried out data extraction, assessed the risk of bias and assessed the quality of the evidence using GRADE.
Main results: We included nine RCTs recruiting 1104 adults and children with SCD (haemoglobin SS (HbSS), haemoglobin SC (HbSC) or haemoglobin Sβºthalassaemia (HbSβºthal) genotypes). Studies lasted from six to 30 months. We judged the quality of the evidence for the first two comparisons below as moderate to low as the studies contributing to these comparisons were mostly large and well-designed (and at low risk of bias); however, the evidence was limited and imprecise for some outcomes such as quality of life, deaths during the studies and adverse events, and the results are applicable only to individuals with HbSS and HbSβºthal genotypes. We judged the quality of the evidence for the third and fourth comparisons to be very low due to the limited number of participants, the lack of statistical power (both studies were terminated early with approximately only 20% of their target sample size recruited) and the lack of applicability to all age groups and genotypes. Hydroxyurea versus placebo Five studies (784 adults and children with HbSS or HbSβºthal) compared hydroxyurea to placebo; four recruited individuals with only severe disease and one recruited individuals with all disease severities. Hydroxyurea probably improves pain alteration (using measures such as pain crisis frequency, duration, intensity, hospital admissions and opoid use) and life-threatening illness, but we found no difference in death rates (10 deaths occurred during the studies, but the rates did not differ by treatment group) (all moderate-quality evidence). Hydroxyurea may improve measures of HbF (low-quality evidence) and probably decreases neutrophil counts (moderate-quality evidence). There were no consistent differences in terms of quality of life and adverse events (including serious or life-threatening events) (low-quality evidence). There were fewer occurrences of acute chest syndrome and blood transfusions in the hydroxyurea groups. Hydroxyurea and phlebotomy versus transfusion and chelation Two studies (254 children with HbSS or HbSβºthal also with risk of primary or secondary stroke) contributed to this comparison. There were no consistent differences in terms of pain alteration, death or adverse events (low-quality evidence) or life-threatening illness (moderate-quality evidence). Hydroxyurea with phlebotomy probably increased HbF and decreased neutrophil counts (moderate-quality evidence), but there were more occurrences of acute chest syndrome and infections. Quality of life was not reported. In the primary prevention study, no strokes occurred in either treatment group but in the secondary prevention study, seven strokes occurred in the hydroxyurea and phlebotomy group (none in the transfusion and chelation group) and the study was terminated early. Hydroxyurea versus observation One study (22 children with HbSS or HbSβºthal also at risk of stoke) compared hydroxyurea to observation. Pain alteration and quality of life were not reported. There were no differences in life-threatening illness, death (no deaths reported in either group) or adverse events (very low-quality evidence). We are uncertain if hydroxyurea improves HbF or decreases neutrophil counts (very low-quality evidence). Treatment regimens with and without hydroxyurea One study (44 adults and children with HbSC) compared treatment regimens with and without hydroxyurea. Pain alteration, life-threatening illness and quality of life were not reported. There were no differences in death rates (no deaths reported in either group), adverse events or neutrophil levels (very low-quality evidence). We are uncertain if hydroxyurea improves HbF (very low-quality evidence).
Authors' conclusions: There is evidence to suggest that hydroxyurea may be effective in decreasing the frequency of pain episodes and other acute complications in adults and children with sickle cell anaemia of HbSS or HbSβºthal genotypes and in preventing life-threatening neurological events in those with sickle cell anaemia at risk of primary stroke by maintaining transcranial Doppler velocities. However, there is still insufficient evidence on the long-term benefits of hydroxyurea, particularly with regard to preventing chronic complications of SCD, or recommending a standard dose or dose escalation to maximum tolerated dose. There is also insufficient evidence about the long-term risks of hydroxyurea, including its effects on fertility and reproduction. Evidence is also limited on the effects of hydroxyurea on individuals with the HbSC genotype. Future studies should be designed to address such uncertainties.
Copyright © 2022 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd.
Conflict of interest statement
Sarah Nevitt: none known.
Angela E Rankine‐Mullings: none known.
Figures
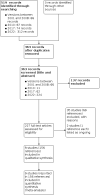
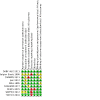













































































Update of
-
Hydroxyurea (hydroxycarbamide) for sickle cell disease.Cochrane Database Syst Rev. 2017 Apr 20;4(4):CD002202. doi: 10.1002/14651858.CD002202.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2022 Sep 1;9:CD002202. doi: 10.1002/14651858.CD002202.pub3. PMID: 28426137 Free PMC article. Updated. Review.
Similar articles
-
Hydroxyurea (hydroxycarbamide) for sickle cell disease.Cochrane Database Syst Rev. 2017 Apr 20;4(4):CD002202. doi: 10.1002/14651858.CD002202.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2022 Sep 1;9:CD002202. doi: 10.1002/14651858.CD002202.pub3. PMID: 28426137 Free PMC article. Updated. Review.
-
Blood transfusion for preventing primary and secondary stroke in people with sickle cell disease.Cochrane Database Syst Rev. 2020 Jul 27;7(7):CD003146. doi: 10.1002/14651858.CD003146.pub4. Cochrane Database Syst Rev. 2020. PMID: 32716555 Free PMC article.
-
Blood transfusion for preventing primary and secondary stroke in people with sickle cell disease.Cochrane Database Syst Rev. 2017 Jan 17;1(1):CD003146. doi: 10.1002/14651858.CD003146.pub3. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2020 Jul 27;7:CD003146. doi: 10.1002/14651858.CD003146.pub4. PMID: 28094851 Free PMC article. Updated. Review.
-
Interventions for preventing silent cerebral infarcts in people with sickle cell disease.Cochrane Database Syst Rev. 2020 Apr 6;4(4):CD012389. doi: 10.1002/14651858.CD012389.pub3. Cochrane Database Syst Rev. 2020. PMID: 32250453 Free PMC article.
-
Interventions for preventing silent cerebral infarcts in people with sickle cell disease.Cochrane Database Syst Rev. 2017 May 13;5(5):CD012389. doi: 10.1002/14651858.CD012389.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2020 Apr 6;4:CD012389. doi: 10.1002/14651858.CD012389.pub3. PMID: 28500860 Free PMC article. Updated. Review.
Cited by
-
A cellular reporter system to evaluate endogenous fetal hemoglobin induction and screen for therapeutic compounds.Hemasphere. 2024 Aug 6;8(8):e139. doi: 10.1002/hem3.139. eCollection 2024 Aug. Hemasphere. 2024. PMID: 39108322 Free PMC article.
-
Effects of hydroxyurea on fertility in male and female sickle cell disease patients. A systemic review and meta-analysis.PLoS One. 2024 Jun 7;19(6):e0304241. doi: 10.1371/journal.pone.0304241. eCollection 2024. PLoS One. 2024. PMID: 38848387 Free PMC article.
-
Transcranial Doppler Ultrasonography-Related Research in the Caribbean Region.Cureus. 2023 Feb 18;15(2):e35147. doi: 10.7759/cureus.35147. eCollection 2023 Feb. Cureus. 2023. PMID: 36949970 Free PMC article. Review.
-
mARC1 Is the Main Contributor to Metabolic Reduction of N-Hydroxyurea.J Med Chem. 2024 Oct 24;67(20):18090-18097. doi: 10.1021/acs.jmedchem.4c01148. Epub 2024 Oct 13. J Med Chem. 2024. PMID: 39397364 Free PMC article.
-
Looking ahead: ethical and social challenges of somatic gene therapy for sickle cell disease in Africa.Gene Ther. 2024 May;31(5-6):202-208. doi: 10.1038/s41434-023-00429-7. Epub 2023 Nov 27. Gene Ther. 2024. PMID: 38012299 Free PMC article. Review.
References
References to studies included in this review
BABY HUG 2011 {published data only}
-
- Adams RJ, Barredo J, Bonds DR, Brown C, Casella J, Daner L, et al. TCD in infants: a report from the BABY HUG trial. Blood 2005;106(11):952. [ABSTRACT NO: 952] [CFGD REGISTER: SC180f]
-
- Adams RJ, Luden J, Miller S, Wang W, Rees R, Li D, et al. TCD in infants: a report from the Baby Hug study. In: 28th Annual Meeting of the National Sickle Cell Disease Program; 2005 Apr 9-13; Cincinnati, Ohio. 2005:105. [CFGD REGISTER: SC180a]
-
- Alvarez O, Miller ST, Wang WC, Luo Z, McCarville MB, Schwartz GJ, et al. Effect of hydroxyurea treatment on renal function parameters: results from the multi-center placebo-controlled BABY HUG clinical trial for infants with sickle cell anemia. Pediatric Blood & Cancer 2012;59(4):668-74. [CENTRAL: 848700] [CFGD REGISTER: SC180gg] [PMID: ] - PMC - PubMed
-
- Armstrong FD, Elkin TD, Brown RC, Glass P, Rees RC, Wang WC, et al. Neurodevelopment in infants with sickle cell anemia: baseline data from the Baby HUG trial. Blood 2008;112(11):713. [CFGD REGISTER: SC180p]
Belgian Study 1996 {published data only}
-
- Ferster A, Vermylen C, Cornu G, Buyse M, Corazza F, Devalck C, et al. Hydroxyurea for treatment of severe sickle cell anemia: a pediatric clinical trial. Blood 1996;88(6):1960-4. - PubMed
CHAMPS 2011 {published data only}
-
- NCT00532883. Hydroxyurea and magnesium pidolate to treat people with hemoglobin sickle cell disease. https://ClinicalTrials.gov/show/NCT00532883 (first received 21 September 2007).
-
- Wang W, Brugnara C, Snyder C, Wynn L, Rogers Z, Kalinyak K, et al. The effects of hydroxycarbamide and magnesium on haemoglobin SC disease: results of the multi-centre CHAMPS trial. British Journal of Haematology 2011;152(6):771-6. [CENTRAL: 801819] [PMID: ] - PubMed
-
- Wang WC, Snyder C, Brugnara C, Telen MJ, Steinberg MH, Wynn LW, et al. Effects of hydroxyurea (HU) and magnesium pidolate (Mg) in hemoglobin SC disease (HbSC): the "CHAMPS" trial. Blood 2009;22. [ABSTRACT NO: 819]
Jain 2012 {published data only}
-
- Jain D. Low dose hydroxyurea in children severely affected with sickle cell disease: hospital based randomized controlled study. American Journal of Hematology 2010;85(8):E42. [DOI: 10.1002/ajh.21763] - DOI
-
- Jain D. Low dose hydroxyurea in children severely affected with sickle cell disease: hospital based randomized controlled study. In: 4th Annual Sickle Cell Disease Research and Educational Symposium & Grant Writing Institute and Annual Sickle Cell Disease Scientific Meeting; 2010 Feb 14-19; Hollywood, Florida. 2010:52. [ABSTRACT NO.: 076] [CENTRAL: CN-00746336] [CFGD REGISTER: SC211b]
-
- Jain DL, Sarathi V, Desai S, Bhatnagar M, Lodha A. Low fixed-dose hydroxyurea in severely affected Indian children with sickle cell disease. Hemoglobin 2012;36(4):323-32. [CENTRAL: 879848] [CFGD REGISTER: SC234] [PMID: ] - PubMed
MSH 1995 {published data only}
-
- Armstrong FD, Steinberg MH, Ballas SK, Ataga KI, Waclawiw MA, Kutlar A, et al. Development outcomes of offspring of adults treated with hydroxyurea in the multicenter study of hydroxyurea. Blood 2009;(22). [ABSTRACT NO: 1543]
-
- Ballas SK, Barton F, Castro O, Bellevue R, Investigators of the multicenter study of hydroxyurea in sickle cell anemia. Narcotic analgesia use among adult patients with sickle cell anemia. Blood 1995;86(10 Suppl 1):642a.
-
- Ballas SK, Barton F, Castro O, Koshy M, Bellevue R. Pattern of narcotic analgesic consumption among adult patients with sickle cell anemia. In: National Sickle Cell Disease Program 21st Annual Meeting; 1996 Mar. 1996:63.
-
- Ballas SK, Bauserman RL, McCarthy WF, Castro OL, Smith WR, Waclawiw MA, Investigators of the Multicenter Study of Hydroxyurea in Sickle Cell Anemia. Hydroxyurea and acute painful crises in sickle cell anemia: effects on hospital length of stay and opioid utilization during hospitalization, outpatient acute care contacts, and at home. Journal of Pain and Symptom Management 2010;40(6):870-82. [CENTRAL: 779191] [PMID: ] - PMC - PubMed
NOHARM 2017 {published data only}
-
- Carman AS, Sautter C, Anyanwu JN, Ssemata AS, Opoka RO, Ware RE, et al. Perceived benefits and risks of participation in a clinical trial for Ugandan children with sickle cell anemia. Pediatric Blood & Cancer 2020;67(2):e27830. [CENTRAL: CN-02080753] [CFGD REGISTER: SC286k] [EMBASE: 628049884] [PMID: ] - PubMed
-
- Dong M, Marahatta A, Opoka RO, McGann PT, John CC, Ware RE, et al. PK/PD modeling of hydroxyurea treatment effects on fetal hemoglobin levels in young African children with sickle cell anemia. Journal of Pharmacokinetics and Pharmacodynamics 2018;45(Suppl 1):S80. [CENTRAL: CN-01653655] [CFGD REGISTER: SC286j] [EMBASE: 624153366]
-
- Marahatta A, Dong M, Opoka R, McElhinney KE, Latham TS, John CC, et al. Pharmacokinetics of hydroxyurea therapy in African children with sickle cell anemia: a NOHARM ancillary PK study. Blood 2017;130(Suppl 1):2252. [CENTRAL: CN-01450314] [CFGD REGISTER: SC286f] [DOI: 10.1182/blood.V130.Suppl_1.2252.2252] [EMBASE: 620385125] - DOI
-
- NCT01976416. Novel use of hydroxyurea in an African region with malaria. www.ClinicalTrials.gov/show/NCT01976416 (first received 5 November 2013).
SCATE 2015 {published data only}
-
- Hankins JS, McCarville MB, Rankine-Mullings A, Reid ME, Lobo CL, Moura PG, et al. Prevention of conversion to abnormal transcranial Doppler with hydroxyurea in sickle cell anemia: a phase III international randomized clinical trial. American Journal of Hematology 2015;90(12):1099-105. [DOI: ] - PMC - PubMed
-
- NCT01531387. Sparing Conversion to Abnormal TCD (Transcranial Doppler) Elevation (SCATE). http://clinicaltrials.gov/show/NCT01531387 (first received 13 February 2012).
SWiTCH 2012 {published data only}
-
- Alvarez O, Yovetich NA, Scott JP, Owen W, Miller ST, Schultz W, et al. Pain and other non-neurological adverse events in children with sickle cell anemia and previous stroke who received hydroxyurea and phlebotomy or chronic transfusions and chelation: results from the SWiTCH clinical trial. American Journal of Hematology 2013;88(11):932-8. [CENTRAL: 963136] [PMID: ] - PMC - PubMed
-
- Aygun B, Mortier NA, Kesler K, Lockhart A, Schultz WH, Cohen AR, et al. Therapeutic phlebotomy is safe in children with sickle cell anaemia and can be effective treatment for transfusional iron overload. British Journal of Haematology 2015;169(2):262-6. [CFGD REGISTER: SC220k] [PMID: ] - PMC - PubMed
-
- Aygun B, Mortier NA, Kesler K, Schultz WH, Alvarez OA, Rogers ZR, et al. Therapeutice phlebotomy in children with sickle cell anemia, stroke, and iron overload: the SWiTCH experience. In: 53rd ASH Annual Meeting and Exposition; 2011 Dec 10-13; San Diego, California. 2011. [ABSTRACT NO: 1044]
-
- Kwiatkowski JL, Cohen AR, Garro J, Alvarez O, Nagasubramanian R, Sarnaik S, et al. Transfusional iron overload in children with sickle cell anemia on chronic transfusion therapy for secondary stroke prevention. American Journal of Hematology 2012;87(2):221-3. [CENTRAL: 864015] [PMID: ] - PubMed
TWiTCH 2016 {published data only}
-
- Aygun B, Wruck LM, Schultz WH, Mueller BU, Brown C, Luchtman-Jones L, et al. Chronic transfusion practices for prevention of primary stroke in children with sickle cell anemia and abnormal TCD velocities. American Journal of Hematology 2012;87(4):428-30. - PubMed
-
- Aygun B, Mortier N, Rogers ZR, Owen W, Fuh B, George A, et al. Iron unloading by therapeutic phlebotomy in previously transfused children with sickle cell anemia: the twitch experience. Blood 2016;128(22):1018. [CFGD REGISTER: SC276i]
-
- De Montalembert M, Benkerrou M, Grosse R, Kordes U, Brousse V, Pondarre C, et al. Assessment of hematological data in a cohort of European children with sickle cell anemia treated with hydroxyurea: can European centers apply today the lessons from the twitch study? Blood 2016;128(22):2494. [CFGD REGISTER: SC276k]
-
- Helton KJ, Roberts D, Schultz WH, Davis BR, Kalfa TA, Pressel SL, et al. Effects of chronic transfusion therapy on MRI and MRA in children with sickle cell anemia. Blood 2014;124(21):4052. [CENTRAL: 1261883]
-
- Imran H, Aygun B, Davis BR, Pressel SL, Herbert Schultz W, Jackson SM, et al. Effects of chronic transfusion therapy on transcranial doppler ultrasonography velocities in children with sickle cell anemia at risk for primary stroke: baseline findings from the Twitch trial. Blood 2014;124(21):87. [CENTRAL: 1261882]
References to studies excluded from this review
Abdullahi 2020a {published data only}
-
- Abdullahi SU, Sunusi SM, Sani Abba M, Sani S, Galadanci A, Inuwa H, et al. Low-versus moderate-dose hydroxyurea for secondary stroke prevention in children with sickle cell disease in sub-Saharan Africa: final results of a randomized controlled trial, sprint trial. Blood 2020;136(Suppl 1):5-6. [CFGD REGISTER: SC280b]
-
- NCT02675790. Low dose hydroxyurea for secondary stroke prevention in children with sickle cell disease in Sub-Saharan Africa (SPRINT). www.clinicaltrials.gov/ct2/show/NCT02675790 (first received 8 January 2016). [CFGD REGISTER: SC280a]
Abdullahi 2020b {published data only}
-
- Abdullahi SU, Jibir BW, Bello-Manga H, Gambo S, Inuwa H, Tijjani AG, et al. Hydroxyurea for primary stroke prevention in children with sickle cell anaemia in Nigeria (SPRING): a double-blind, multicentre, randomised, phase 3 trial. Lancet. Haematology 2022;9(1):e26-e37. [CFGD REGISTER: SC293e] - PMC - PubMed
-
- Abdullahi SU, Jibir BW, Bello-Manga H, Gambo S, Inuwa H, Tijjani AG, et al. Randomized controlled trial of fixed low- vs moderate-dose hydroxyurea for primary stroke prevention in sub-Saharan Africa: final results of the spring trial. Blood 2020;136(Suppl 1):4-5. [CFGD REGISTER: SC293d]
-
- Abdullahi SU, Wudil BJ, Bello-Manga H, Musa AB, Gambo S, Galadanci NA, et al. Primary prevention of stroke in children with sickle cell anemia in sub-Saharan Africa: rationale and design of phase III randomized clinical trial. Pediatric Hematology and Oncology 2021;38(1):49-64. [CFGD REGISTER: SC293b] - PMC - PubMed
-
- Bello-Manga H, Galadanci AA, Abdullahi S, Ali S, Jibir B, Gambo S, et al. Low educational level of head of household, as a proxy for poverty, is associated with severe anaemia among children with sickle cell disease living in a low-resource setting: evidence from the SPRING trial. British Journal of Haematology 2020;190(6):939-44. [CFGD REGISTER: SC293c] - PMC - PubMed
Al‐Nood 2011 {published data only}
-
- Al-Nood HA, Al-Khawlani MM, Al-Akwa A. Fetal hemoglobin response to hydroxyurea in Yemeni sickle cell disease patients. Hemoglobin 2011;35(1):13-21. - PubMed
Charnigo 2019 {published data only}
-
- Charnigo RJ, Beidler D, Rybin D, Pittman DD, Tan B, Howard J, et al. PF-04447943, a phosphodiesterase 9A inhibitor, in stable sickle cell disease patients: a phase Ib randomized, placebo-controlled study. Clinical and Translational Science 2019;12(2):180-8. [CENTRAL: CN-01937374] [CFGD REGISTER: SC345b] [EMBASE: 626983905] [PMID: ] - PMC - PubMed
-
- Charnigo RJ, Howard J, Beidler D, Rybin D, Tan B, Michelson AD, et al. A phase 1b, randomized, double-blind, placebo-controlled study of PF-04447943 in patients with stable sickle cell disease: changes in exploratory biomarkers. Blood 2017;130(Suppl 1):974. [CENTRAL: CN-01616847] [CFGD REGISTER: SC345a]
-
- NCT02114203. Safety, tolerability, pharmacokinetics, and pharmacodynamics study of PF-04447943, co-administered with and without hydroxyurea, in subjects with stable sickle cell disease [A phase 1b, randomized, double-blind (sponsor open), placebo controlled study to evaluate the safety, tolerability, pharmacokinetics, and pharmacodynamics of pf 04447943, co-administered with and without hydroxyurea, in subjects with stable sickle cell disease]. www.clinicaltrials.gov/show/NCT02114203 (first received 15 April 2014).
Conran 2019 {published data only}
-
- Conran N, Fertrin KY, Maximo CDA, Oliveira T, Lobo C, Costa FF. Safe use of hydroxyurea in sickle cell disease patients hospitalized for painful vaso-occlusive episodes: results of the randomized, open-label helps study. Blood 2019;134:2303. [CFGD REGISTER: SC431b] - PubMed
-
- NCT03062501. Hydroxyurea in the emergency room to lessen pain in sickle cell crisis. www.ClinicalTrials.gov/show/NCT03062501 (first received 23 February 2017). [CFGD REGISTER: SC431a]
CTRI/2022/01/039317 {published data only}
-
- CTRI/2022/01/039317. Low dose hydroxyurea in sickle cell anemia [Efficacy, safety, and population pharmacokinetics of low-dose vs. standard dose hydroxyurea in paediatric patients suffering from sickle cell disease: a randomized double-blind active-control non-inferiority clinical trial]. trialsearch.who.int/Trial2.aspx?TrialID=CTRI/2022/01/039317 (first received 27 January 2022).
De Montalembert 2006 {published data only}
-
- De Montalembert M, Bachir D, Hulin A, Gimeno L, Mogenet A, Bresson JL, et al. A phase 1 pharmacokinetics (PK) study of hydroxyurea (HU) 1,000 mg coated breakable tablets and 500mg capsules in pediatric and adult patients with sickle cell disease. Blood 2005;106(11). [ABSTRACT NO: 3194] [CFGD REGISTER: SC193a] [DOI: 10.1182/blood.V106.11.3194.3194] - DOI - PubMed
-
- Montalembert M, Bachir D, Hulin A, Gimeno L, Mogenet A, Bresson JL, et al. Pharmacokinetics of hydroxyurea 1,000 mg coated breakable tablets and 500 mg capsules in pediatric and adult patients with sickle cell disease. Haematologica 2006;91(12):1685-8. - PubMed
de Oliveira 2019 {published data only}
Eleuterio 2019 {published data only}
-
- Eleuterio RM, Nascimento FO, Araujo TG, Castro MF, Filho TPA, Filho PA, et al. Double-blind clinical trial of arginine supplementation in the treatment of adult patients with sickle cell anaemia. Advances in Hematology 2019;eCollection 2019:4397150. [CENTRAL: CN-01916183] [CFGD REGISTER: SC383] [DOI: 10.1155/2019/4397150] [EMBASE: 626558583] - DOI - PMC - PubMed
Estepp 2016 {published data only}
-
- Estepp JH, Melloni C, Thornburg CD, Wiczling P, Rogers Z, Rothman JA, et al. Pharmacokinetics and bioequivalence of a liquid formulation of hydroxyurea in children with sickle cell anemia. Journal of Clinical Pharmacology 2016;56(3):298-306. [CENTRAL: CN-01137410] [CFGD REGISTER: SC393] [EMBASE: 607382494] - PMC - PubMed
Field 2020 {published data only}
-
- NCT01960413. Phase 2 study of montelukast for the treatment of sickle cell anemia. www.clinicaltrials.gov/show/NCT01960413 (first received 10 October 2013). [CFGD REGISTER: SC294a]
Meier 2020 {published data only}
-
- NCT03789591. Hydroxyurea optimization through precision study (HOPS). clinicaltrials.gov/show/nct03789591 (first received 28 Dec 2018). [CFGD REGISTER: SC430a]
Misra 2017 {published data only}
-
- NCT01848925. A phase I open-label, randomized, safety and efficacy study of SANGUINATE™ at two doses levels versus hydroxyurea in sickle cell disease (SCD) patients. www.clinicaltrials.gov/show/NCT01848925 (first received 8 May 2013). [CFGD REGISTER: SC290a]
NCT00000602 {published data only}
-
- NCT00000602. Pediatric hydroxyurea in sickle cell anemia (PED HUG). www.ClinicalTrials.gov/show/NCT00000602 (first received 28 October 1999).
NCT00001197 {published data only}
-
- NCT00001197. Hydroxyurea for the treatment of patients with sickle cell anemia. www.ClinicalTrials.gov/show/NCT00001197 (first received 4 November 1999).
NCT00004492 {published data only}
-
- NCT00004492. Phase I/II randomized study of hydroxyurea with or without clotrimazole in patients with sickle cell anemia. www.clinicaltrials.gov/show/NCT00004492 (first received 19 October 1999).
NCT00672789 {published data only}
-
- NCT00672789. Adherence to hydroxyurea in children with sickle cell anemia. clinicaltrials.gov/show/NCT00672789 (first received 6 May 2008). [CFGD REGISTER: SC422]
NCT00890396 {published data only}
-
- NCT00890396. Long- term effects of hydroxyurea in children with sickle cell anemia (the BABY HUG follow-up study). clinicaltrials.gov/ct2/show/NCT00890396 (first received 29 April 2009).
NCT02090296 {published data only}
-
- NCT02090296. Risk-based therapy for sickle cell anemia: a feasibility study. www.ClinicalTrials.gov/show/NCT02090296 (first received 18 March 2014). [CFGD REGISTER: SC291]
NCT02149537 {published data only}
-
- NCT02149537. Risk stratification for clinical severity of sickle cell disease in Nigeria and assessment of efficacy and safety during treatment with hydroxyurea. www.clinicaltrials.gov/show/NCT02149537 (first received 24 May 2014). [CFGD REGISTER: SC289]
NCT03634488 {published data only}
-
- NCT03634488. Management of severe acute malnutrition in SCD, in Northern Nigeria. www.ClinicalTrials.gov/show/NCT03634488 (first received 16 August 2018).
NCT03825341 {published data only}
-
- NCT03825341. Hydroxyurea therapy: optimizing access in pediatric populations everywhere. clinicaltrials.gov/show/NCT03825341 (first received 31 January 2019). [CFGD REGISTER: SC432]
NCT04675645 {published data only}
-
- NCT04675645. Adherence to HU and HRQOL in patients with sickle cell disease: an intervention study using hu-go app. clinicaltrials.gov/show/NCT04675645 (first received 19 December 2020). [CFGD REGISTER: SC415]
NCT05142254 {published data only}
-
- A trial for prevention of recurrent ischemic priapism in men with sickle cell disease: a pilot study (PIN). www.clinicaltrials.gov/ct2/show/NCT05142254 (first received 2 December 2021).
-
- PACTR202105561969346. A randomized controlled double-blind trial for prevention of recurrent ischemic priapism in men with sickle cell disease: a pilot study. Priapism in Nigeria (PIN) trial. www.who.int/trialsearch/Trial2.aspx?TrialID=PACTR202105561969346 2021.
NDEPTH 2013 {published data only}
-
- George A, Aygun B, Mortier N, Sparreboom A, Ware R. A randomized controlled trial of a dose-prediction equation to determine maximum tolerated dose of hydroxyurea in children with sickle cell anemia. Pediatric Blood & Cancer 2013;60(Suppl). [ABSTRACT NO: 588] [CENTRAL: 1007869] [CFGD REGISTER: SC283b] [EMBASE: 71047876]
-
- George A, Dinu B, Estrada N, Minard C, Hurwitz RL, Mahoney D, et al. NDEPTH: a randomized controlled trial of a novel dose-prediction equation to determine maximum tolerated dose for hydroxyurea therapy in pediatric patients with sickle cell anemia. Blood 2019;134:2267. [CENTRAL: CN-02050621] [CFGD REGISTER: SC283c] [EMBASE: 630318481]
-
- George A, Dinu B, Estrada N, Minard CG, Hurwitz R, Mahoney DH, et al. Novel dose escalation to predict treatment with hydroxyurea (NDEPTH): a randomized controlled trial of a dose-prediction equation to determine maximum tolerated dose of hydroxyurea in pediatric sickle cell disease. American Journal of Hematology 2020;95(9):E242-4. [CENTRAL: CN-02131373] [CFGD REGISTER: SC283d] [EMBASE: 631937641] [PMID: ] - PubMed
-
- George A, Dinu BR, Ware RE. Ndepth: novel dose escalation to predict treatment with hydroxyurea. Blood 2015;126(23):3419. [CFGD REGISTER: SC283a]
NOHARM 2020 {published data only}
-
- John CC, Opoka R, Latham T, Hume H, Nakafeero M, Kasirye P, et al. Optimizing hydroxyurea dosing in sickle cell anemia: the Uganda MTD study. Blood 2019;134:520. [CENTRAL: CN-02051498] [CFGD REGISTER: SC435b] [EMBASE: 630317942]
-
- John CC, Opoka RO, Latham TS, Hume HA, Nabaggala C, Kasirye P, et al. Hydroxyurea dose escalation for sickle cell anemia in Sub-Saharan Africa. New England Journal of Medicine 2020;382(26):2524-33. [CENTRAL: CN-02133119] [CFGD REGISTER: SC435c] [PMID: ] - PubMed
-
- NCT03128515. Optimizing hydroxyurea therapy in children with SCA in malaria endemic areas [Optimizing hydroxyurea therapy in children with sickle cell anemia in malaria endemic areas: the NOHARM maximum tolerated dose (MTD) study]. www.clinicaltrials.gov/show/NCT03128515 (first received 25 April 2017). [CFGD REGISTER: SC435a]
Pushi 2000 {published data only}
-
- Pushi A. Hydroxyurea ameliorates importantly the clinical course of sickle cell disease reducing the frequency of painful crises. Hematology Journal 2000;1(Suppl 1):34. [ABSTRACT NO.: 130] [CFGD REGISTER: SC282]
Silva‐Pinto 2007 {published data only}
-
- Silva-Pinto AC, Carrara RC, Oliveira VC, Palma PV, Campos AD, Zago MA, et al. Hydroxyurea treatment of sickle cell diseases causes megaloblastic transformation of the bone marrow that is responsible for the increase of the mean corpuscular volume. Haematologica 2007;Suppl 1:298. [CFGD REGISTER: SC201]
Silva‐Pinto 2014 {published data only}
-
- Silva-Pinto AC, Dias-Carlos C, Saldanha-Araujo F, Ferreira FI, Palma PV, Araujo AG, et al. Hydroxycarbamide modulates components involved in the regulation of adenosine levels in blood cells from sickle-cell anemia patients. Annals of Hematology 2014;93(9):1457-65. [CENTRAL: 1000846] [CFGDREGISTER: SC259] [JID:: 9107334] [PMID: ] - PubMed
Smith 2022 {published data only}
-
- McClish DK, Smith WR, Okhomiuna V, Mouaffo DS, Lottenberg R, Chen I, et al. The association of painful crises with patient reported outcomes in sickle cell disease: the SHIP-HU study. Blood 2018;132(Suppl 1):3509. [CFGD REGISTER: SC413a] [DOI: 10.1182/blood-2018-99-114869] - DOI
-
- Smith WR, McClish DK, Johnson S, Lottenberg R, Sisler I, Sop D, et al. The effect of patient navigators on health-related quality of life in sickle cell anemia: the SHIP-HU study. Blood 2019;134 Suppl 1:2168. [CFGD REGISTER: SC413c]
-
- Smith WR, McClish DK, Lottenberg R, Sisler I, Sop D, Johnson S, et al. The effect of patient navigators on laboratory parameters of hydroxyurea adherence in sickle cell anemia: the ship-hu study. Blood 2019;134 Suppl 1:2309. [CFGD REGISTER: SC413b]
-
- Smith WR, McClish DK, Lottenberg R, Sisler IY, Sop D, Johnson S, et al. A randomised controlled provider-blinded trial of community health workers in sickle cell anaemia: effects on haematologic variables and hydroxyurea adherence. British Journal of Haematology 2022;196(1):193-203. [CFGD REGISTER: SC413d] - PubMed
STAND 2019 {published data only}
-
- Abboud MR, Howard J, Cancado R, Smith WR, Guvenc B, Espurz N, et al. Crizanlizumab versus placebo, with or without hydroxyurea/hydroxycarbamide, in adolescent and adult patients with sickle cell disease and vaso-occlusive crises: a randomized, double-blind, phase III study (STAND). Blood 2019;134:998. [CENTRAL: CN-02048780] [CFGD REGISTER: SC392] [EMBASE: 630316641]
STEADFAST 2019 {published data only}
-
- Ataga KI, Saraf SL, Derebail VK, Sharpe CC, Inati A, Lebensburger JD, et al. The effect of crizanlizumab plus standard of care (Soc) versus soc alone on renal function in patients with sickle cell disease and chronic kidney disease: a randomized, multicenter, open-label, phase ii study (Steadfast). Blood 2019;134:1018. [CENTRAL: CN-02050662] [CFGD REGISTER: SC391a] [EMBASE: 630319168]
-
- EUCTR2018-003608-38-GR. Study exploring the effect of crizanlizumab on kidney function in patients with chronic kidney disease caused by sickle cell disease [A phase II, multicenter, randomized, open label two arm study comparing the effect of crizanlizumab + standard of care to standard of care alone on renal function in sickle cell disease patients = 16 years with chronic kidney disease due to sickle cell nephropathy (STEADFAST) - STEADFAST]. www.who.int/trialsearch/Trial2.aspx?TrialID=EUCTR2018-003608-38-GR (first received 2019). [CENTRAL: CN-02068279] [CFGD REGISTER: SC391b]
SUSTAIN 2019 {published data only}
-
- Ataga KI, Kutlar A, Cancado R, Liles D, Velez-Nandayapa L, Lincy J, et al. Crizanlizumab treatment is not associated with the development of proteinuria and hematuria in patients with sickle cell disease: a safety analysis from the sustain study. Hemasphere 2018;2(S1):305-6. [CFGD REGISTER: SC287h]
-
- Ataga KI, Kutlar A, DeBonnett L, Lincy J, Kanter J. Crizanlizumab treatment is associated with clinically significant reductions in hospitalization in patients with sickle cell disease: results from the sustain study. Blood 2019;134:2289. [CENTRAL: CN-02052376] [CFGD REGISTER: SC287j] [EMBASE: 630317661]
-
- Ataga KI, Kutlar A, Kanter J, Liles D, Cancado R, Friedrisch J, et al. SUSTAIN: a multicenter, randomized, placebo-controlled, double-blind, 12-month study to assess safety and efficacy of selg1 with or without hydroxyurea therapy in sickle cell disease patients with sickle cell-related pain crises. Blood 2016;128(22):1. [CFGD REGISTER: SC287f]
-
- Kanter J, Kutlar A, Liles D, Cancado R, Bruederle A, Shi M, et al. Crizanlizumab 5.0 mg/kg increased the time to first on-treatment sickle cell pain crisis: a subgroup analysis of the phase II sustain study. Blood 2017;130(Suppl 1):613. [CFGD REGISTER: SC287c]
Vichinsky 2013 {published data only}
-
- Vichinsky E, Torres M, Glass J, Minniti CP, Barrette S, Habr D, et al. A randomized phase II study evaluating the efficacy and safety of deferasirox versus deferoxamine in patients with sickle cell disease (SCD): 2-year results including pharmacokinetics (PK) and safety of deferasirox with concomitant hydroxyurea therapy. Blood 2011;118(21):1082. [ABSTRACT NO: 1082] [CFGD REGISTER: SC233a]
-
- Vichinsky E, Torres M, Minniti CP, Barrette S, Habr D, Zhang Y, et al. Efficacy and safety of deferasirox compared with deferoxamine in sickle cell disease: two-year results including pharmacokinetics and concomitant hydroxyurea. American Journal of Hematology 2013;88(12):1068-73. [CFGD REGISTER: SC233b] [PMID: ] - PubMed
Voskaridou 2005 {published data only}
-
- Voskaridou E, Terpos E, Margeli A, Hantzi E, Meletis J, Papassotiriou I, et al. Renal dysfunction and osteodystrophy in patients with sickle cell thalassaemia under long-term treatment with hydroxyurea. In: 10th Congress of the European Hematology Association; 2005 June 2-5; Stockholm International Fairs, Sweden. 2005. [ABSTRACT NO: 0174] [CFGD REGISTER: SC168]
References to ongoing studies
NCT01389024 {published data only}
-
- NCT01389024. Hydroxyurea to prevent brain injury in sickle cell disease (HUPrevent). www.clinicaltrials.gov/ct2/show/NCT01389024 (first received 30 June 2011).
NCT03806452 {published data only}
-
- NCT03806452. SIKAMIC (SIklos on Kidney Function and AlbuMInuria Clinical Trial). www.clinicaltrials.gov/ct2/show/NCT03806452 (first received 16 January 2019).
Additional references
Anyanwu 2016
Aumont 2015
-
- Aumont C, Driss F, Lazure T, Picard V, Creidy R, De Botton S, et al. Myelodysplastic syndrome with clonal cytogenetic abnormalities followed by fatal erythroid leukemia after 14 years of exposure to hydroxyurea for sickle cell anemia. American Journal of Hematology 2015;90(7):E131-2. - PubMed
Ballas 1996
-
- Ballas SK, Barton F, Castro O, Koshy M, Bellevue R. Pattern of narcotic analgesic consumption among adult patients with sickle cell anemia. In: National Sickle Cell Disease Program 21st Annual Meeting; 1996 Mar. 1996:63.
Ballas 2006
Ballas 2009
-
- Ballas SK, Guo N, DeCastro L, Bellevue R. Exposure to hydroxyurea and pregnancy outcomes in patients with sickle cell anemia. JAMA 2009;101(10):1046. - PubMed
Barton 1996
-
- Barton F, Terrin M, Moore R, McMahon RP, Charache S. Ascertainment of the primary end point in the Multicenter Study of Hydroxyrea in Sickle Cell Anemia (MSH). The MSH Investigators. Controlled Clinical Trials 1996;17:67S. - PubMed
Baz 2012
Berk 1995
-
- Berk PD, Wasserman LR, Fruchtman SM, Goldberg JD. Treatment of polycythemia vera: a summary of clinical trials conducted by the Polycythemia Vera Study Group. In: Wasserman LR BP, Berlin NI, editors(s). Polycythemia Vera and the Myeloproliferation Disorders. Philadelphia: WB Saunders, 1995:166-94.
Bernaudin 2016
-
- Bernaudin F, Verlhac S, Arnaud C, Kamdem A, Hau I, Leveillé E, et al. Long-term treatment follow-up of children with sickle cell disease monitored with abnormal transcranial Doppler velocities. Blood 2016;127(15):1814-22. [DOI: 10.1182/blood-2015-10-675231. Epub 2016 Feb 5.] - PubMed
Berthaut 2008
-
- Berthaut I, Guignedoux G, Kirsch-Noir F, Larouziere V, Ravel C, Bachir D, et al. Influence of sickle cell disease and treatment with hydroxyurea on sperm parameters and fertility of human males. Haematologica 2008;93(7):988-93. - PubMed
Brousseau 2010
-
- Brousseau DC, A Panepinto J, Nimmer M, Hoffmann RG. The number of people with sickle‐cell disease in the United States: national and state estimates. American Journal of Hematology 2010;85(1):77-8. - PubMed
Brozovic 1987
Chakravorty 2015
Charache 1995
-
- Charache S, Terrin TL, Moore RD, Dover GJ, Barton FB, Eckert SV, et al. Effect of hydroxyurea on the frequency of painful crises in sickle cell anaemia. New England Journal of Medicine 1995;332:1317-22. - PubMed
Davies 1997a
-
- Davies SC, Roberts-Harewood M. Blood transfusion in sickle cell disease. Blood Reviews 1997;11:57-71. - PubMed
Davies 1997b
Elbourne 2002
-
- Elbourne DR, Altman DG, Higgins JPT, Curtin F, Worthington HV, Vail A. Meta-analysis involving cross-over trials: methodological issues. International Journal of Epidemiology 2002;31(1):140-9. - PubMed
Fruchtman 1994
-
- Fruchtman SM, Kaplan ME, Peterson P, Mack K, Berk PD, Wasserman LR. Acute leukemia (AL), hydroxyurea (HU), and polycythemia vera (PV): an analysis of risk from the Polycythemia Vera Study Group. Blood 1994;84(Suppl 1):518a.
Fuggle 1996
Gardner 2016
-
- Gardner K, Douiri A, Drasar E, Allman M, Mwirigi A, Awogbade M, et al. Survival in adults with sickle cell disease in a high income setting. Blood 2016;128(10):1436-8. - PubMed
Gray 1991
Green 2014
Grosse 2011
Handy 1996
-
- Handy C, Barton F, Moore R, McMahon R, Eckert S, Terrin M. Dose titration in the Multicentre Study of Hydroxyurea in sickle cell anemia (MSH). Controlled Clinical Trials 1996;17(Suppl 2):92S.
Hankins 2014
Hassell 2010
-
- Hassell KL. Population estimates of sickle cell disease in the US. American Journal of Preventive Medicine 2010;38(4):S512-21. - PubMed
Higgins 2003
Higgins 2011
-
- Higgins JPT, Altman DG, Sterne JAC (editors). Chapter 8: Assessing risk of bias in included studies. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from www.cochrane-handbook.org.
Howard 2015
-
- Howard J, Telfer P. Sickle Cell Disease in Clinical Practice. London: Springer-Verlag, 2015.
Jones 2009
-
- Jones AP, Riley RD, Williamson PR, Whitehead A. Meta-analysis of individual patient data versus aggregate data from longitudinal clinical trials. Clinical Trials 2009;6(1):16-27. - PubMed
King 2015
-
- King LG, Bortolusso-Ali S, Cunningham-Myrie CA, Reid ME. Impact of a Comprehensive Sickle Cell Center on early childhood mortality in a developing country: the Jamaican experience. Journal of Pediatrics 2015;167(3):702-5. - PubMed
Lanzkron 2013
Luchtman‐Jones 2016
-
- Luchtman‐Jones L, Pressel S, Hilliard L, Brown RC, Smith MG, Thompson AA, et al. Effects of hydroxyurea treatment for patients with hemoglobin SC disease. American Journal of Hematology 2016;91(2):238-42. - PubMed
Manci 2003
-
- Manci EA, Culberson DE, Yang YM, Gardner TM, Powell R, Haynes J Jr, et al. Causes of death in sickle cell disease: an autopsy study. British Journal of Haematology 2003;123(2):359-65. - PubMed
McGann 2011
Najean 1997
-
- Najean Y, Rain JD. Treatment of polycythemia vera: the use of hydroxyurea and pipobroman in 292 patients under the age of 65 years. Blood 1997;90(9):3370-7. - PubMed
National Toxicology Program 2008
-
- National Toxicology Program. NTP-CERHR monograph on the potential human reproductive and developmental effects of hydroxyurea. NTP CERHR MON 2008;Oct(21):vii-viii, v, ix-III1. [NIH PUBLICATION NO: 08-5993] - PubMed
NCEPOD 2008
-
- NCEPOD. ‘Sickle: A sickle crisis?’ A report of the national confidential enquiry into patient outcome and death. 2008. www.ncepod.org.uk.2008sc.html (accessed 19 September 2016).
NICE 2012
-
- National Institute of Clinical Excellence (NICE). Sickle cell acute painful episode: management of an acute painful sickle cell episode in hospital. www.nice.org.uk/guidance/cg143 (accessed 6 March 2017) 2012. - PubMed
NIH 2014
-
- Evidence Based Management of Sickle Cell Disease. Expert Panel Report. National Institute of Health. 2014. www.nhlbi.nih.gov/sites/www.nhlbi.nih.gov/files/sickle-cell-disease-repo... (accessed 19 September 2016).
Perrine 1978
-
- Perrine RP, Pembrey ME, John P, Perrine S, Shoup F. Natural history of sickle cell anaemia in Saudi Arabs: a study of 270 subjects. Annals of Internal Medicine 1978;88:1-6. - PubMed
Perronne 2002
-
- Perronne V, Roberts-Harewood M, Bachir D, Roudot-Thoraval F, Delord JM, Thuret I, et al. Patterns of mortality in sickle cell disease in adults in France and England. Haematology Journal 2002;3(1):56-60. - PubMed
Piel 2013
Platt 1984
-
- Platt OS, Orkin SH, Dover GJ, Beardsley GP, Miller B, Nathan DG. Hydroxyurea increases fetal hemoglobin production in sickle cell anemia. Transactions of the Association of American Physicians 1984;97:268-74. - PubMed
Platt 1994
-
- Platt OS, Brambilla DJ, Rosse WF, Milner PF, Castro O, Steinberg MH, et al. Mortality in sickle cell disease - life expectancy and risk factors for early death. New England Journal of Medicine 1994;330:1639-43. - PubMed
Powars 1983
-
- Powars D, Overturf G, Turner E. Is there an increased risk of Haemophilus influenzae septicemia in sickle cell anaemia? Pediatrics 1983;71:927-31. - PubMed
Quinn 2010
Rankine‐Mullings 2020
-
- Rankine-Mullings AE, Logan TM, King L-G, Cunningham-Myrie CA, Scott CR, Knight-Madden JM. The risk of acute events among patients with sickle cell disease in relation to early or late initiation of care at a specialist center: evidence from a retrospective cohort study. BMC Pediatrics 2020;20(1):373. - PMC - PubMed
Rogers 1978
Serjeant 2018
Steinberg 1999
-
- Steinberg MH. The management of sickle cell disease. New England Journal of Medicine 1999;340:1021-30. - PubMed
Steinberg 2003
-
- Steinberg MH, Barton F, Castro O, Pegelow CH, Ballas SK, Kutlar A, et al. Effect of hydroxyurea on mortality and morbidity in adult sickle cell anemia: risks and benefits up to 9 years of treatment. JAMA 2003;289(13):1645-51. - PubMed
Steinberg 2010
Sterkers 1998
-
- Sterkers Y, Preudhomme C, Lai JL, Demory JL, Caulier MT, Wattel E, et al. Acute myeloid leukemia and myelodysplastic syndromes following essential thrombocythemia treated with hydroxyurea: high proportion of cases with 17p deletion. Blood 1998;91(2):616-22. - PubMed
STOP 2006
Strouse 2008
-
- Strouse JJ, Lanzkron S, Beach MC, Haywood C, Park H, Witkop C, et al. Hydroxyurea for sickle cell disease: a systematic review for efficacy and toxicity in children. Pediatrics 2008;122(6):1332-43. - PubMed
Telfer 2007
-
- Telfer P, Coen P, Chakravorty S, Wilkey O, Evans J, Newell H, et al. Clinical outcomes in children with sickle cell disease living in England: a neonatal cohort in East London. Haematologica 2007;92(7):905-12. - PubMed
Terrin 1999
-
- Terrin ML, Barton FB, Bonds D, Ballas SK, Swerdlow P, Pegelow CH, et al. Effect of hydroxyurea on quality of life: 2-year results from the multicenter study of hydroxyurea in sickle cell anemia. In: National Sickle Cell Disease Program 23rd Annual Meeting; 1999 Mar. 1999:161.
Triadou 1994
-
- Triadou P, Maier-Redelsperger M, Krishnamoorty R, Deschamps A, Casadevall N, Dunda O, et al. Fetal haemoglobin variations following hydroxyurea treatment in patients with cyanotic congenital heart disease. Nouvelle Revue Francaise d Hematologie 1994;36(5):367-72. - PubMed
van der Plas 2011
-
- Plas EM, van den Tweel XW, Geskus RB, Heijboer H, Biemond BJ, Peters M, et al. Mortality and causes of death in children with sickle cell disease in the Netherlands, before the introduction of neonatal screening. British Journal of Haematology 2011;155(1):106-10. - PubMed
Veith 1985
-
- Veith R, Galanello R, Papayannopoulou T, Stamatoyannopoulos G. Stimulation of F-cell production in patients with sickle cell anaemia treated with cytarabine or hydroxyurea. New England Journal of Medicine 1985;313:1571-5. - PubMed
Voskaridou 2010
-
- Voskaridou E, Christoulas D, Bilalis A, Plata E, Varvagiannis K, Stamatopoulos G, et al. The effect of prolonged administration of hydroxyurea on morbidity and mortality in adult patients with sickle cell syndromes: results of a 17-year, single-centre trial (LaSHS). Blood 2010;115:2354-63. - PubMed
Ware 2010
Wierenga 2001
-
- Wierenga KJ, Hambleton IR, Lewis NA. Survival estimates for patients with homozygous sickle-cell disease in Jamaica: a clinic-based population study. Lancet 2001;357(9257):680-3. - PubMed
Zimmerman 2004
-
- Zimmerman SA, Schultz WH, Davis JS, Pickens CV, Mortier NA, Howard TA, et al. Sustained long-term hematologic efficacy of hydroxyurea at maximum tolerated dose in children with sickle cell disease. Blood 2004;103(6):2039-45. - PubMed
References to other published versions of this review
Jones 2001
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

