Enfortumab Vedotin Plus Pembrolizumab in Previously Untreated Advanced Urothelial Cancer
- PMID: 36041086
- PMCID: PMC10476837
- DOI: 10.1200/JCO.22.01643
Enfortumab Vedotin Plus Pembrolizumab in Previously Untreated Advanced Urothelial Cancer
Abstract
Purpose: Cisplatin-based combination chemotherapy remains the standard of care for locally advanced or metastatic urothelial cancer (la/mUC); however, toxicity is substantial, responses are rarely durable, and many patients with la/mUC are ineligible. Each enfortumab vedotin and pembrolizumab have shown a survival benefit versus chemotherapy in UC, are not restricted by cisplatin eligibility, and warrant investigation as a first-line (1L) combination therapy in patients ineligible for cisplatin.
Methods: In this ongoing phase Ib/II, multicenter, open-label study, 1L cisplatin-ineligible patients with la/mUC received enfortumab vedotin 1.25 mg/kg once daily on days 1 and 8 and pembrolizumab 200 mg (day 1) intravenously once daily in 3-week cycles. The primary end point was safety. Key secondary end points included confirmed objective response rate, duration of response (DOR), and overall survival (OS).
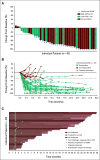
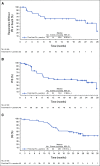
Results: Forty-five patients received enfortumab vedotin plus pembrolizumab. The most common treatment-related adverse events (TRAEs) were peripheral sensory neuropathy (55.6%), fatigue (51.1%), and alopecia (48.9%). Twenty-nine patients (64.4%) had grade 3 or higher TRAEs; the most common were increased lipase (17.8%), maculopapular rash (11.1%), and fatigue (11.1%). One death (2.2%) was classified as a TRAE. The confirmed objective response rate after a median of nine cycles was 73.3% with a complete response rate of 15.6%. The median DOR and median OS were 25.6 months and 26.1 months, respectively.
Conclusion: Enfortumab vedotin plus pembrolizumab showed a manageable safety profile. Most patients experienced tumor shrinkage. The median DOR and median OS exceeding 2 years in a cisplatin-ineligible patient population make this a promising combination currently under investigation in a phase III study (ClinicalTrials.gov identifier: NCT04223856).
Trial registration: ClinicalTrials.gov NCT03288545 NCT04223856.
Conflict of interest statement
Enfortumab Vedotin Plus Pembrolizumab in Previously Untreated Advanced Urothelial Cancer
The following represents disclosure information provided by authors of this manuscript. All relationships are considered compensated unless otherwise noted. Relationships are self-held unless noted. I = Immediate Family Member, Inst = My Institution. Relationships may not relate to the subject matter of this manuscript. For more information about ASCO's conflict of interest policy, please refer to
Open Payments is a public database containing information reported by companies about payments made to US-licensed physicians (
Figures
Similar articles
-
Enfortumab Vedotin With or Without Pembrolizumab in Cisplatin-Ineligible Patients With Previously Untreated Locally Advanced or Metastatic Urothelial Cancer.J Clin Oncol. 2023 Sep 1;41(25):4107-4117. doi: 10.1200/JCO.22.02887. Epub 2023 Jun 27. J Clin Oncol. 2023. PMID: 37369081 Free PMC article. Clinical Trial.
-
Enfortumab Vedotin and Pembrolizumab in Untreated Advanced Urothelial Cancer.N Engl J Med. 2024 Mar 7;390(10):875-888. doi: 10.1056/NEJMoa2312117. N Engl J Med. 2024. PMID: 38446675 Clinical Trial.
-
Enfortumab vedotin after PD-1 or PD-L1 inhibitors in cisplatin-ineligible patients with advanced urothelial carcinoma (EV‑201): a multicentre, single-arm, phase 2 trial.Lancet Oncol. 2021 Jun;22(6):872-882. doi: 10.1016/S1470-2045(21)00094-2. Epub 2021 May 12. Lancet Oncol. 2021. PMID: 33991512 Clinical Trial.
-
Managing potential adverse events during treatment with enfortumab vedotin + pembrolizumab in patients with advanced urothelial cancer.Front Oncol. 2024 Apr 22;14:1326715. doi: 10.3389/fonc.2024.1326715. eCollection 2024. Front Oncol. 2024. PMID: 38711854 Free PMC article. Review.
-
The evolving treatment landscape of metastatic urothelial cancer.Nat Rev Urol. 2024 Oct;21(10):580-592. doi: 10.1038/s41585-024-00872-0. Epub 2024 May 3. Nat Rev Urol. 2024. PMID: 38702396 Review.
Cited by
-
Impact of DNA Repair Deficiency in the Evolving Treatment Landscape of Bladder Cancer.Curr Urol Rep. 2024 Oct 9;26(1):12. doi: 10.1007/s11934-024-01242-4. Curr Urol Rep. 2024. PMID: 39382743 Review.
-
Efficacy and safety of the first-line systemic treatments in patients with advanced-stage urothelial carcinoma: a systematic review and network meta-analysis.Front Oncol. 2024 Sep 16;14:1468784. doi: 10.3389/fonc.2024.1468784. eCollection 2024. Front Oncol. 2024. PMID: 39351347 Free PMC article.
-
Pre-first-line chemotherapy risk stratification for overall survival in advanced urothelial carcinoma in sequential therapy era.World J Urol. 2024 May 9;42(1):307. doi: 10.1007/s00345-024-04952-z. World J Urol. 2024. PMID: 38722418
-
Bladder cancer.Nat Rev Dis Primers. 2023 Oct 26;9(1):58. doi: 10.1038/s41572-023-00468-9. Nat Rev Dis Primers. 2023. PMID: 37884563 Free PMC article. Review.
-
Novel bispecific antibody-drug conjugate targeting PD-L1 and B7-H3 enhances antitumor efficacy and promotes immune-mediated antitumor responses.J Immunother Cancer. 2024 Oct 2;12(10):e009710. doi: 10.1136/jitc-2024-009710. J Immunother Cancer. 2024. PMID: 39357981 Free PMC article.
References
-
- Dash A, Galsky MD, Vickers AJ, et al. : Impact of renal impairment on eligibility for adjuvant cisplatin-based chemotherapy in patients with urothelial carcinoma of the bladder. Cancer 107:506-513, 2006 - PubMed
-
- Galsky MD, Hahn NM, Rosenberg J, et al. : Treatment of patients with metastatic urothelial cancer "unfit" for cisplatin-based chemotherapy. J Clin Oncol 29:2432-2438, 2011 - PubMed
-
- Sternberg CN, de Mulder P, Schornagel JH, et al. : Seven year update of an EORTC phase III trial of high-dose intensity M-VAC chemotherapy and G-CSF versus classic M-VAC in advanced urothelial tract tumours. Eur J Cancer 42:50-54, 2006 - PubMed

