The Primary Microglial Leukodystrophies: A Review
- PMID: 35683020
- PMCID: PMC9181167
- DOI: 10.3390/ijms23116341
The Primary Microglial Leukodystrophies: A Review
Abstract
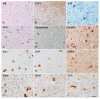
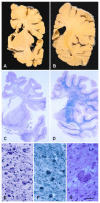
Primary microglial leukodystrophy or leukoencephalopathy are disorders in which a genetic defect linked to microglia causes cerebral white matter damage. Pigmented orthochromatic leukodystrophy, adult-onset orthochromatic leukodystrophy associated with pigmented macrophages, hereditary diffuse leukoencephalopathy with (axonal) spheroids, and adult-onset leukoencephalopathy with axonal spheroids and pigmented glia (ALSP) are different terms apparently used to designate the same disease. However, ALSP linked to dominantly inherited mutations in CSF1R (colony stimulating factor receptor 1) cause CSF-1R-related leukoencephalopathy (CRP). Yet, recessive ALSP with ovarian failure linked to AARS2 (alanyl-transfer (t)RNA synthase 2) mutations (LKENP) is a mitochondrial disease and not a primary microglial leukoencephalopathy. Polycystic membranous lipomembranous osteodysplasia with sclerosing leukoencephalopathy (PLOSL; Nasu-Hakola disease: NHD) is a systemic disease affecting bones, cerebral white matter, selected grey nuclei, and adipose tissue The disease is caused by mutations of one of the two genes TYROBP or TREM2, identified as PLOSL1 and PLOSL2, respectively. TYROBP associates with receptors expressed in NK cells, B and T lymphocytes, dendritic cells, monocytes, macrophages, and microglia. TREM2 encodes the protein TREM2 (triggering receptor expressed on myeloid cells 2), which forms a receptor signalling complex with TYROBP in macrophages and dendritic cells. Rather than pure microglial leukoencephalopathy, NHD can be considered a multisystemic "immunological" disease.
Keywords: AARS; CRP; CSFR1; LKENP; Nasu–Hakola disease; TREM2; TYROBP; adult-onset (or dominant) leukodystrophy with axonal spheroids and pigmented glia; adult-onset orthochromatic leukodystrophy associated with pigmented macrophages; hereditary diffuse leukoencephalopathy with axonal spheroids; pigmented orthochromatic leukodystrophy; polycystic membranous lipomembranous osteodysplasia with sclerosing leukoencephalopathy.
Conflict of interest statement
The author declares no conflict of interest.
Figures

Similar articles
-
Adult onset leukoencephalopathy with axonal spheroids and pigmented glia (ALSP) and Nasu-Hakola disease: lesion staging and dynamic changes of axons and microglial subsets.Brain Pathol. 2017 Nov;27(6):748-769. doi: 10.1111/bpa.12443. Epub 2017 Mar 2. Brain Pathol. 2017. PMID: 27608278 Free PMC article.
-
Early-onset dementia, leukoencephalopathy and brain calcifications: a clinical, imaging and pathological comparison of ALSP and PLOSL/Nasu Hakola disease.Acta Neurol Belg. 2018 Dec;118(4):607-615. doi: 10.1007/s13760-018-1023-8. Epub 2018 Sep 21. Acta Neurol Belg. 2018. PMID: 30242731
-
Analysis of Mutations in AARS2 in a Series of CSF1R-Negative Patients With Adult-Onset Leukoencephalopathy With Axonal Spheroids and Pigmented Glia.JAMA Neurol. 2016 Dec 1;73(12):1433-1439. doi: 10.1001/jamaneurol.2016.2229. JAMA Neurol. 2016. PMID: 27749956
-
Adult-onset leukoencephalopathy with axonal spheroids and pigmented glia (ALSP): Integrating the literature on hereditary diffuse leukoencephalopathy with spheroids (HDLS) and pigmentary orthochromatic leukodystrophy (POLD).J Clin Neurosci. 2018 Feb;48:42-49. doi: 10.1016/j.jocn.2017.10.060. Epub 2017 Nov 6. J Clin Neurosci. 2018. PMID: 29122458 Review.
-
Adult-onset leukoencephalopathy with axonal spheroids and pigmented glia.Handb Clin Neurol. 2024;204:263-271. doi: 10.1016/B978-0-323-99209-1.00005-3. Handb Clin Neurol. 2024. PMID: 39322383 Review.
Cited by
-
Deciphering glial contributions to CSF1R-related disorder via single-nuclear transcriptomic profiling: a case study.Acta Neuropathol Commun. 2024 Aug 28;12(1):139. doi: 10.1186/s40478-024-01853-5. Acta Neuropathol Commun. 2024. PMID: 39217398 Free PMC article.
-
Primary ovarian insufficiency: update on clinical and genetic findings.Front Endocrinol (Lausanne). 2024 Sep 26;15:1464803. doi: 10.3389/fendo.2024.1464803. eCollection 2024. Front Endocrinol (Lausanne). 2024. PMID: 39391877 Free PMC article. Review.
-
Advancing cell therapy for neurodegenerative diseases.Cell Stem Cell. 2023 May 4;30(5):512-529. doi: 10.1016/j.stem.2023.03.017. Epub 2023 Apr 20. Cell Stem Cell. 2023. PMID: 37084729 Free PMC article. Review.
-
TREM2 as an evolving therapeutic target in Alzheimer's disease.Neural Regen Res. 2023 Dec;18(12):2680-2681. doi: 10.4103/1673-5374.371360. Neural Regen Res. 2023. PMID: 37449620 Free PMC article. No abstract available.
-
Defects in lysosomal function and lipid metabolism in human microglia harboring a TREM2 loss of function mutation.Acta Neuropathol. 2023 Jun;145(6):749-772. doi: 10.1007/s00401-023-02568-y. Epub 2023 Apr 28. Acta Neuropathol. 2023. PMID: 37115208 Free PMC article.
References
Publication types
MeSH terms
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

