Surfactin Shows Relatively Low Antimicrobial Activity against Bacillus subtilis and Other Bacterial Model Organisms in the Absence of Synergistic Metabolites
- PMID: 35456828
- PMCID: PMC9030240
- DOI: 10.3390/microorganisms10040779
Surfactin Shows Relatively Low Antimicrobial Activity against Bacillus subtilis and Other Bacterial Model Organisms in the Absence of Synergistic Metabolites
Abstract
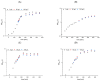
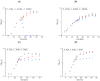
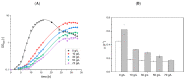
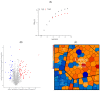
Surfactin is described as a powerful biosurfactant and is natively produced by Bacillus subtilis in notable quantities. Among other industrially relevant characteristics, antimicrobial properties have been attributed to surfactin-producing Bacillus isolates. To investigate this property, stress approaches were carried out with biotechnologically established strains of Corynebacterium glutamicum, Bacillus subtilis, Escherichia coli and Pseudomonas putida with the highest possible amounts of surfactin. Contrary to the popular opinion, the highest growth-reducing effects were detectable in B. subtilis and E. coli after surfactin treatment of 100 g/L with 35 and 33%, respectively, while P. putida showed no growth-specific response. In contrast, other antimicrobial biosurfactants, like rhamnolipids and sophorolipids, showed significantly stronger effects on bacterial growth. Since the addition of high amounts of surfactin in defined mineral salt medium reduced the cell growth of B. subtilis by about 40%, the initial stress response at the protein level was analyzed by mass spectrometry, showing induction of stress proteins under control of alternative sigma factors σB and σW as well as the activation of LiaRS two-component system. Overall, although surfactin is associated with antimicrobial properties, relatively low growth-reducing effects could be demonstrated after the surfactin addition, challenging the general claim of the antimicrobial properties of surfactin.
Keywords: Bacillus subtilis; biosurfactants; secondary metabolites; stress response; surfactin.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Corner Flows Induced by Surfactant-Producing Bacteria Bacillus subtilis and Pseudomonas fluorescens.Microbiol Spectr. 2022 Oct 26;10(5):e0323322. doi: 10.1128/spectrum.03233-22. Epub 2022 Oct 10. Microbiol Spectr. 2022. PMID: 36214703 Free PMC article.
-
Co-production of surfactin and a novel bacteriocin by Bacillus subtilis subsp. subtilis H4 isolated from Bikalga, an African alkaline Hibiscus sabdariffa seed fermented condiment.Int J Food Microbiol. 2013 Apr 1;162(3):297-307. doi: 10.1016/j.ijfoodmicro.2013.01.013. Epub 2013 Jan 29. Int J Food Microbiol. 2013. PMID: 23466466
-
Stress-Responsive Alternative Sigma Factor SigB Plays a Positive Role in the Antifungal Proficiency of Bacillus subtilis.Appl Environ Microbiol. 2019 Apr 18;85(9):e00178-19. doi: 10.1128/AEM.00178-19. Print 2019 May 1. Appl Environ Microbiol. 2019. PMID: 30824454 Free PMC article.
-
Production of microbial biosurfactants: Status quo of rhamnolipid and surfactin towards large-scale production.Biotechnol J. 2017 Jul;12(7). doi: 10.1002/biot.201600561. Epub 2017 May 23. Biotechnol J. 2017. PMID: 28544628 Review.
-
Bacillus licheniformis: The unexplored alternative for the anaerobic production of lipopeptide biosurfactants?Biotechnol Adv. 2022 Nov;60:108013. doi: 10.1016/j.biotechadv.2022.108013. Epub 2022 Jun 22. Biotechnol Adv. 2022. PMID: 35752271 Review.
Cited by
-
A Family of Cyclic Lipopeptides Found in Human Isolates of Bacillus Ameliorates Acute Colitis via Direct Agonism of Toll-Like Receptor 2 in a Murine Model of Inflammatory Bowel Disease.Dig Dis Sci. 2024 Oct;69(10):3729-3741. doi: 10.1007/s10620-024-08534-2. Epub 2024 Aug 7. Dig Dis Sci. 2024. PMID: 39110366 Free PMC article.
-
Evaluation of an external foam column for in situ product removal in aerated surfactin production processes.Front Bioeng Biotechnol. 2023 Nov 6;11:1264787. doi: 10.3389/fbioe.2023.1264787. eCollection 2023. Front Bioeng Biotechnol. 2023. PMID: 38026897 Free PMC article.
-
Microbial Biosurfactant as an Alternate to Chemical Surfactants for Application in Cosmetics Industries in Personal and Skin Care Products: A Critical Review.Biomed Res Int. 2023 Apr 13;2023:2375223. doi: 10.1155/2023/2375223. eCollection 2023. Biomed Res Int. 2023. PMID: 37090190 Free PMC article. Review.
-
Improving Surfactin Production in Bacillus subtilis 168 by Metabolic Engineering.Microorganisms. 2024 May 15;12(5):998. doi: 10.3390/microorganisms12050998. Microorganisms. 2024. PMID: 38792827 Free PMC article.
-
History of a model plant growth-promoting rhizobacterium, Bacillus velezensis GB03: from isolation to commercialization.Front Plant Sci. 2023 Oct 11;14:1279896. doi: 10.3389/fpls.2023.1279896. eCollection 2023. Front Plant Sci. 2023. PMID: 37885658 Free PMC article. Review.
References
-
- Bartal A., Vigneshwari A., Bóka B., Vörös M., Takács I., Kredics L., Manczinger L., Varga M., Vágvölgyi C., Szekeres A. Effects of Different Cultivation Parameters on the Production of Surfactin Variants by a Bacillus subtilis Strain. Molecules. 2018;23:2675. doi: 10.3390/molecules23102675. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

