Effective GH Replacement With Once-weekly Somapacitan vs Daily GH in Children with GHD: 3-year Results From REAL 3
- PMID: 34964458
- PMCID: PMC9016428
- DOI: 10.1210/clinem/dgab928
Effective GH Replacement With Once-weekly Somapacitan vs Daily GH in Children with GHD: 3-year Results From REAL 3
Abstract
Context: Current GH therapy requires daily injections, which can be burdensome. Somapacitan is a long-acting GH derivative in development for treatment of GH deficiency (GHD).
Objective: Evaluate the efficacy, safety, and tolerability of once-weekly somapacitan after 3 years of treatment.
Design: A multicenter, randomized, controlled, phase 2 study comparing somapacitan and once-daily GH for 156 weeks (NCT02616562).
Setting: Twenty-nine sites in 11 countries.
Patients: Fifty-nine children with GHD randomized (1:1:1:1) and exposed to treatment. Fifty-three children completed the 3-year period.
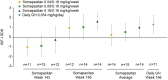
Interventions: Patients received somapacitan (0.04 [n = 14], 0.08 [n = 15], or 0.16 [n = 14] mg/kg/wk) or daily GH (n = 14) (0.034 mg/kg/d, equivalent to 0.238 mg/kg/wk) subcutaneously during the first year, after which all patients on somapacitan received 0.16 mg/kg/wk.
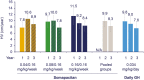
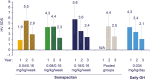
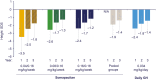
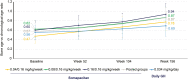
Main outcome measures: Height velocity (HV) at year 3; changes from baseline in height SD score (HSDS), HVSDS, and IGF-I SDS.
Results: The estimated treatment difference (95% CI) in HV for somapacitan 0.16/0.16 mg/kg/wk vs daily GH at year 3 was 0.8 cm/y (-0.4 to 2.1). Change in HVSDS from baseline to year 3 was comparable between somapacitan 0.16/0.16 mg/kg/wk, the pooled somapacitan groups, and daily GH. A gradual increase in HSDS from baseline was observed for all groups. At year 3, mean HSDS was similar for the pooled somapacitan groups and daily GH. Change from baseline to year 3 in mean IGF-I SDS was similar across treatments.
Conclusions: Once-weekly somapacitan in children with GHD showed sustained efficacy over 3 years in all assessed height-based outcomes with similar safety and tolerability to daily GH. A plain language summary (1) is available for this study.
Clinical trial information: This study has been registered at ClinicalTrials.gov, number NCT02616562 (REAL 3).
Keywords: childhood growth hormone deficiency; growth hormone; growth hormone deficiency; growth hormone replacement therapy; long-acting growth hormone; somapacitan.
© The Author(s) 2021. Published by Oxford University Press on behalf of the Endocrine Society.
Figures


Similar articles
-
Once-Weekly Somapacitan vs Daily GH in Children With GH Deficiency: Results From a Randomized Phase 2 Trial.J Clin Endocrinol Metab. 2020 Apr 1;105(4):e1847-61. doi: 10.1210/clinem/dgz310. J Clin Endocrinol Metab. 2020. PMID: 31917835 Free PMC article. Clinical Trial.
-
Effective GH Replacement With Somapacitan in Children With GHD: REAL4 2-year Results and After Switch From Daily GH.J Clin Endocrinol Metab. 2023 Nov 17;108(12):3090-3099. doi: 10.1210/clinem/dgad394. J Clin Endocrinol Metab. 2023. PMID: 37406251 Free PMC article. Clinical Trial.
-
Effective growth hormone replacement with once-weekly somapacitan in Japanese children with growth hormone deficiency: Results from REAL4, a phase 3 clinical trial.Clin Endocrinol (Oxf). 2024 Apr;100(4):389-398. doi: 10.1111/cen.15025. Epub 2024 Feb 18. Clin Endocrinol (Oxf). 2024. PMID: 38368603 Clinical Trial.
-
Developments in the Management of Growth Hormone Deficiency: Clinical Utility of Somapacitan.Drug Des Devel Ther. 2024 Feb 3;18:291-306. doi: 10.2147/DDDT.S315172. eCollection 2024. Drug Des Devel Ther. 2024. PMID: 38333899 Free PMC article. Review.
-
Once-Weekly Somapacitan as an Alternative Management of Growth Hormone Deficiency in Prepubertal Children: A Systematic Review and Meta-Analysis of Randomized Controlled Trial.Children (Basel). 2024 Feb 9;11(2):227. doi: 10.3390/children11020227. Children (Basel). 2024. PMID: 38397339 Free PMC article. Review.
Cited by
-
Long acting growth hormone (LAGH), an update.Front Pediatr. 2023 Sep 28;11:1254231. doi: 10.3389/fped.2023.1254231. eCollection 2023. Front Pediatr. 2023. PMID: 37842029 Free PMC article. Review.
-
Weekly Somapacitan is Effective and Well Tolerated in Children With GH Deficiency: The Randomized Phase 3 REAL4 Trial.J Clin Endocrinol Metab. 2022 Nov 25;107(12):3378-3388. doi: 10.1210/clinem/dgac513. J Clin Endocrinol Metab. 2022. PMID: 36062966 Free PMC article. Clinical Trial.
-
Effects of Taekwondo Training on Growth Factors in Normal Korean Children and Adolescents: A Systematic Review and Meta-Analysis of Randomized Controlled Trials.Children (Basel). 2023 Feb 8;10(2):326. doi: 10.3390/children10020326. Children (Basel). 2023. PMID: 36832454 Free PMC article. Review.
-
Long-acting growth hormone in 2022.Pediatr Investig. 2023 Jan 3;7(1):36-42. doi: 10.1002/ped4.12358. eCollection 2023 Mar. Pediatr Investig. 2023. PMID: 36967745 Free PMC article. Review.
-
What do we do now that the long-acting growth hormone is here?Front Endocrinol (Lausanne). 2022 Aug 22;13:980979. doi: 10.3389/fendo.2022.980979. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 36072938 Free PMC article. Review.
References
-
- Sävendahl L. Plain language summary - Effective growth hormone replacement with once-weekly somapacitan versus daily growth hormone in children with growth hormone deficiency: 3-year results from REAL 3. figshare. 2022. 10.6084/m9.figshare.17086649.v1 - DOI
-
- Grimberg A, DiVall SA, Polychronakos C, et al. . Guidelines for growth hormone and insulin-like growth factor-I treatment in children and adolescents: growth hormone deficiency, idiopathic short stature, and primary insulin-like growth factor-I deficiency. Horm. Res Paed. 2016;86(6):361-397. - PubMed
-
- Backeljauw P, Cappa M, Kiess W, et al. . Impact of short stature on quality of life: a systematic literature review. Growth Horm IGF Res. 2021;57-58:101392. - PubMed
-
- Høybye C, Cohen P, Hoffman AR, et al. . Status of long-acting-growth hormone preparations--2015. Growth Horm IGF Res. 2015;25(5):201-206. - PubMed

