Characterization of a Cobalt-Substituted Globin-Coupled Oxygen Sensor Histidine Kinase from Anaeromyxobacter sp. Fw109-5: Insights into Catalytic Regulation by Its Heme Coordination Structure
- PMID: 34963974
- PMCID: PMC8697598
- DOI: 10.1021/acsomega.1c05564
Characterization of a Cobalt-Substituted Globin-Coupled Oxygen Sensor Histidine Kinase from Anaeromyxobacter sp. Fw109-5: Insights into Catalytic Regulation by Its Heme Coordination Structure
Abstract
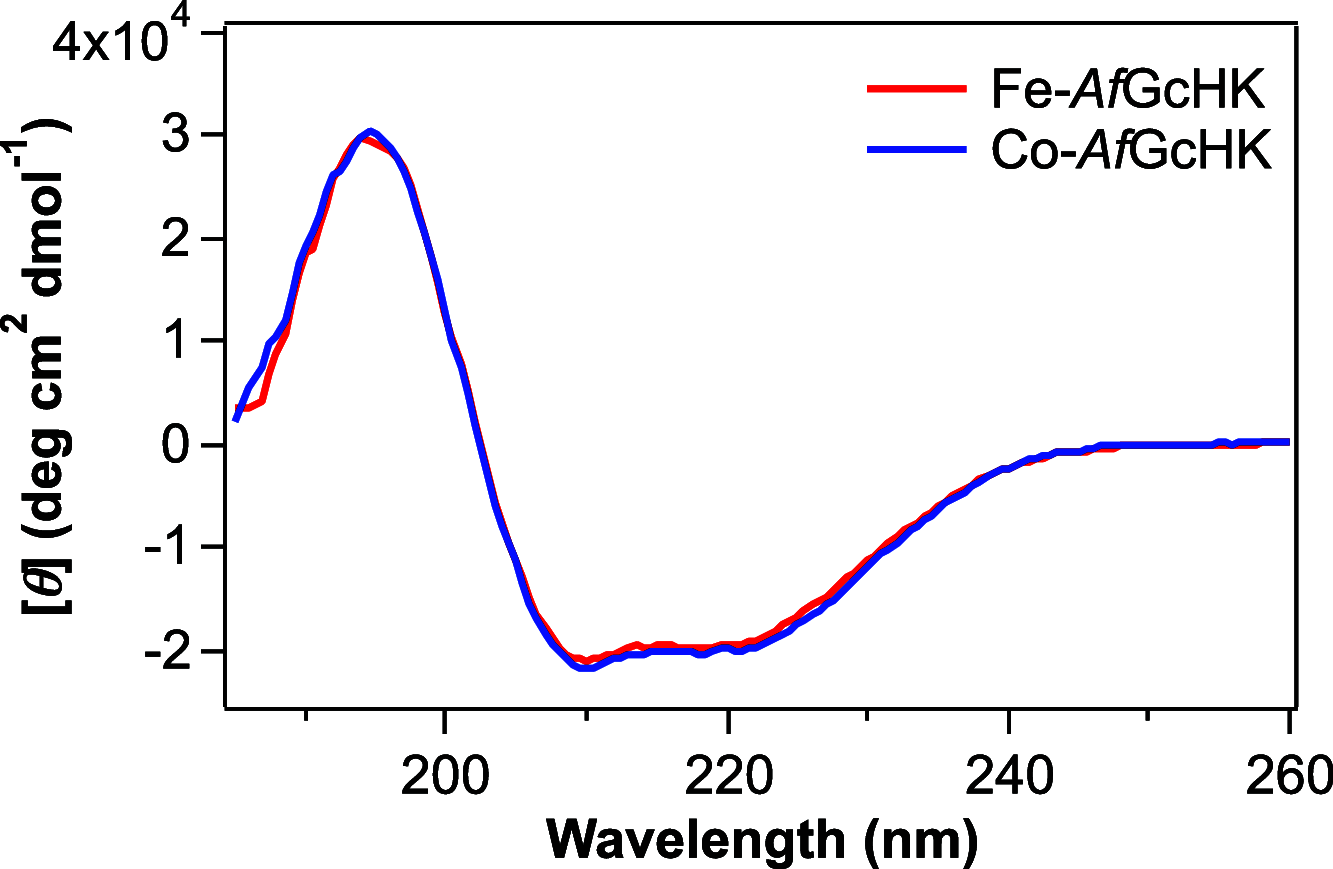
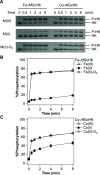
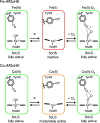
Heme-based gas sensors are an emerging class of heme proteins. AfGcHK, a globin-coupled histidine kinase from Anaeromyxobacter sp. Fw109-5, is an oxygen sensor enzyme in which oxygen binding to Fe(II) heme in the globin sensor domain substantially enhances its autophosphorylation activity. Here, we reconstituted AfGcHK with cobalt protoporphyrin IX (Co-AfGcHK) in place of heme (Fe-AfGcHK) and characterized the spectral and catalytic properties of the full-length proteins. Spectroscopic analyses indicated that Co(III) and Co(II)-O2 complexes were in a 6-coordinated low-spin state in Co-AfGcHK, like Fe(III) and Fe(II)-O2 complexes of Fe-AfGcHK. Although both Fe(II) and Co(II) complexes were in a 5-coordinated state, Fe(II) and Co(II) complexes were in high-spin and low-spin states, respectively. The autophosphorylation activity of Co(III) and Co(II)-O2 complexes of Co-AfGcHK was fully active, whereas that of the Co(II) complex was moderately active. This contrasts with Fe-AfGcHK, where Fe(III) and Fe(II)-O2 complexes were fully active and the Fe(II) complex was inactive. Collectively, activity data and coordination structures of Fe-AfGcHK and Co-AfGcHK indicate that all fully active forms were in a 6-coordinated low-spin state, whereas the inactive form was in a 5-coordinated high-spin state. The 5-coordinated low-spin complex was moderately active-a novel finding of this study. These results suggest that the catalytic activity of AfGcHK is regulated by its heme coordination structure, especially the spin state of its heme iron. Our study presents the first successful preparation and characterization of a cobalt-substituted globin-coupled oxygen sensor enzyme and may lead to a better understanding of the molecular mechanisms of catalytic regulation in this family.
© 2021 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

Similar articles
-
Kinetic Analysis of a Globin-Coupled Histidine Kinase, AfGcHK: Effects of the Heme Iron Complex, Response Regulator, and Metal Cations on Autophosphorylation Activity.Biochemistry. 2015 Aug 18;54(32):5017-29. doi: 10.1021/acs.biochem.5b00517. Epub 2015 Aug 5. Biochemistry. 2015. PMID: 26212354
-
Effects of hydrogen sulfide on the heme coordination structure and catalytic activity of the globin-coupled oxygen sensor AfGcHK.Biometals. 2016 Aug;29(4):715-29. doi: 10.1007/s10534-016-9947-z. Epub 2016 Jul 9. Biometals. 2016. PMID: 27395436
-
Coordination and redox state-dependent structural changes of the heme-based oxygen sensor AfGcHK associated with intraprotein signal transduction.J Biol Chem. 2017 Dec 22;292(51):20921-20935. doi: 10.1074/jbc.M117.817023. Epub 2017 Nov 1. J Biol Chem. 2017. PMID: 29092908 Free PMC article.
-
Signal transduction mechanisms in heme-based globin-coupled oxygen sensors with a focus on a histidine kinase (AfGcHK) and a diguanylate cyclase (YddV or EcDosC).Biol Chem. 2022 Sep 27;403(11-12):1031-1042. doi: 10.1515/hsz-2022-0185. Print 2022 Nov 25. Biol Chem. 2022. PMID: 36165459 Review.
-
Heme-based globin-coupled oxygen sensors: linking oxygen binding to functional regulation of diguanylate cyclase, histidine kinase, and methyl-accepting chemotaxis.J Biol Chem. 2013 Sep 27;288(39):27702-11. doi: 10.1074/jbc.R113.473249. Epub 2013 Aug 8. J Biol Chem. 2013. PMID: 23928310 Free PMC article. Review.
Cited by
-
Applications of the Whole-Cell System in the Efficient Biosynthesis of Heme.Int J Mol Sci. 2023 May 7;24(9):8384. doi: 10.3390/ijms24098384. Int J Mol Sci. 2023. PMID: 37176091 Free PMC article. Review.
References
-
- Antonini E.; Brunori M.. Hemoglobin and Myoglobin in Their Reactions with Ligands; North-Holland Publishing Co.: Amsterdam, 1971.
-
- Igarashi J.; Kitanishi K.; Shimizu T.. Emerging Roles of Heme as a Signal and a Gas-Sensing Site: Heme-Sensing and Gas-Sensing Proteins. In Handbook of Porphyrin Science; Kadish K. M.; Smith K. M.; Guilard R., Eds.; World Scientific Publishing: Hackensack, NJ, 2011; Vol. 15, pp 399–460.
LinkOut - more resources
Full Text Sources

