Hereditary Multiple Exostoses-A Review of the Molecular Background, Diagnostics, and Potential Therapeutic Strategies
- PMID: 34956317
- PMCID: PMC8704583
- DOI: 10.3389/fgene.2021.759129
Hereditary Multiple Exostoses-A Review of the Molecular Background, Diagnostics, and Potential Therapeutic Strategies
Abstract
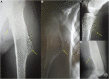
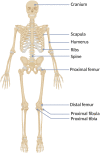
Hereditary multiple exostoses (HMEs) syndrome, also known as multiple osteochondromas, represents a rare and severe human skeletal disorder. The disease is characterized by multiple benign cartilage-capped bony outgrowths, termed exostoses or osteochondromas, that locate most commonly in the juxta-epiphyseal portions of long bones. Affected individuals usually complain of persistent pain caused by the pressure on neighboring tissues, disturbance of blood circulation, or rarely by spinal cord compression. However, the most severe complication of this condition is malignant transformation into chondrosarcoma, occurring in up to 3.9% of HMEs patients. The disease results mainly from heterozygous loss-of-function alterations in the EXT1 or EXT2 genes, encoding Golgi-associated glycosyltransferases, responsible for heparan sulfate biosynthesis. Some of the patients with HMEs do not carry pathogenic variants in those genes, hence the presence of somatic mutations, deep intronic variants, or another genes/loci is suggested. This review presents the systematic analysis of current cellular and molecular concepts of HMEs along with clinical characteristics, clinical and molecular diagnostic methods, differential diagnosis, and potential treatment options.
Keywords: EXT1 gene; EXT2 gene; HME diagnostics; HME molecular backround; HME therapeutic strategies; diaphyseal aclasis; hereditary multiple exostoses (HME); multiple osteochondromas (MO).
Copyright © 2021 Bukowska-Olech, Trzebiatowska, Czech, Drzymała, Frąk, Klarowski, Kłusek, Szwajkowska and Jamsheer.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Mutational screening of EXT1 and EXT2 genes in Polish patients with hereditary multiple exostoses.J Appl Genet. 2014 May;55(2):183-8. doi: 10.1007/s13353-014-0195-z. Epub 2014 Feb 15. J Appl Genet. 2014. PMID: 24532482 Free PMC article.
-
Hereditary multiple exostoses and enchondromatosis.Best Pract Res Clin Rheumatol. 2008 Mar;22(1):45-54. doi: 10.1016/j.berh.2007.12.004. Best Pract Res Clin Rheumatol. 2008. PMID: 18328980 Review.
-
A novel mutation in ext2 caused hereditary multiple exostoses through reducing the synthesis of heparan sulfate.Genet Mol Biol. 2021 May 21;44(2):e20200334. doi: 10.1590/1678-4685-GMB-2020-0334. eCollection 2021. Genet Mol Biol. 2021. PMID: 34042151 Free PMC article.
-
The pathogenic roles of heparan sulfate deficiency in hereditary multiple exostoses.Matrix Biol. 2018 Oct;71-72:28-39. doi: 10.1016/j.matbio.2017.12.011. Epub 2017 Dec 24. Matrix Biol. 2018. PMID: 29277722 Free PMC article. Review.
-
An optimized DHPLC protocol for molecular testing of the EXT1 and EXT2 genes in hereditary multiple osteochondromas.Clin Genet. 2005 Dec;68(6):542-7. doi: 10.1111/j.1399-0004.2005.00538.x. Clin Genet. 2005. PMID: 16283885
Cited by
-
Hereditary Multiple Exostoses with Rare Ocular Finding: A Case Report.J Curr Ophthalmol. 2024 Aug 10;35(4):411-414. doi: 10.4103/joco.joco_128_23. eCollection 2023 Oct-Dec. J Curr Ophthalmol. 2024. PMID: 39281398 Free PMC article.
-
Clinical survey of a pedigree with hereditary multiple exostoses and identification of EXT‑2 gene deletion mutation.Mol Med Rep. 2022 Apr;25(4):141. doi: 10.3892/mmr.2022.12657. Epub 2022 Feb 25. Mol Med Rep. 2022. PMID: 35211766 Free PMC article.
-
Generation of Ext1 Gene-Edited Mice Model Via Dual sgRNAs/Cas9 System and Phenotypic Analyses.Mol Biotechnol. 2024 Nov 20. doi: 10.1007/s12033-024-01325-0. Online ahead of print. Mol Biotechnol. 2024. PMID: 39565542
-
Hereditary multiple exostoses caused by a chromosomal inversion removing part of EXT1 gene.Mol Cytogenet. 2023 May 22;16(1):8. doi: 10.1186/s13039-023-00638-0. Mol Cytogenet. 2023. PMID: 37217936 Free PMC article.
-
Prenatal Diagnosis by Trio Clinical Exome Sequencing: Single Center Experience.Curr Issues Mol Biol. 2024 Apr 6;46(4):3209-3217. doi: 10.3390/cimb46040201. Curr Issues Mol Biol. 2024. PMID: 38666931 Free PMC article.
References
-
- Alabdullrahman L. W., Byerly D. W. (2021). Osteochondroma. Treasure Island (FL): StatPearls Publishing. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

