Use of Yeast Mannoproteins by Oenococcus oeni during Malolactic Fermentation under Different Oenological Conditions
- PMID: 34359413
- PMCID: PMC8305826
- DOI: 10.3390/foods10071540
Use of Yeast Mannoproteins by Oenococcus oeni during Malolactic Fermentation under Different Oenological Conditions
Abstract
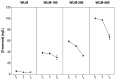
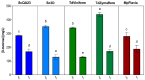
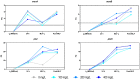
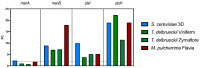
Oenococcus oeni is the main agent of malolactic fermentation in wine. This fermentation takes place after alcoholic fermentation, in a low nutrient medium where ethanol and other inhibitor compounds are present. In addition, some yeast-derived compounds such as mannoproteins can be stimulatory for O. oeni. The mannoprotein concentration in wine depends on the fermenting yeasts, and non-Saccharomyces in particular can increase it. As a result of the hydrolytic activity of O. oeni, these macromolecules can be degraded, and the released mannose can be taken up and used as an energy source by the bacterium. Here we look at mannoprotein consumption and the expression of four O. oeni genes related to mannose uptake (manA, manB, ptsI, and ptsH) in a wine-like medium supplemented with mannoproteins and in natural wines fermented with different yeasts. We observe a general gene upregulation in response to wine-like conditions and different consumption patterns in the studied media. O. oeni was able to consume mannoproteins in all the wines. This consumption was notably higher in natural wines, especially in T. delbrueckii and S. cerevisiae 3D wines, which presented the highest mannoprotein levels. Regardless of the general upregulation, it seems that mannoprotein degradation is more closely related to the fermenting medium.
Keywords: Oenococcus oeni; gene expression; malolactic fermentation; mannoproteins; non-Saccharomyces.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Simulated lees of different yeast species modify the performance of malolactic fermentation by Oenococcus oeni in wine-like medium.Food Microbiol. 2021 Oct;99:103839. doi: 10.1016/j.fm.2021.103839. Epub 2021 May 28. Food Microbiol. 2021. PMID: 34119090
-
Evaluating the effect of using non-Saccharomyces on Oenococcus oeni and wine malolactic fermentation.Food Res Int. 2020 Dec;138(Pt B):109779. doi: 10.1016/j.foodres.2020.109779. Epub 2020 Oct 15. Food Res Int. 2020. PMID: 33288165
-
Molecular adaptation response of Oenococcus oeni in non-Saccharomyces fermented wines: A comparative multi-omics approach.Int J Food Microbiol. 2022 Feb 2;362:109490. doi: 10.1016/j.ijfoodmicro.2021.109490. Epub 2021 Nov 20. Int J Food Microbiol. 2022. PMID: 34844030
-
Non-Saccharomyces in Wine: Effect Upon Oenococcus oeni and Malolactic Fermentation.Front Microbiol. 2018 Mar 23;9:534. doi: 10.3389/fmicb.2018.00534. eCollection 2018. Front Microbiol. 2018. PMID: 29628914 Free PMC article. Review.
-
The use of Torulaspora delbrueckii to improve malolactic fermentation.Microb Biotechnol. 2024 Jan;17(1):e14302. doi: 10.1111/1751-7915.14302. Epub 2023 Jun 30. Microb Biotechnol. 2024. PMID: 37387409 Free PMC article. Review.
Cited by
-
Metabolic Fingerprinting of Muscat of Alexandria Grape Musts during Industrial Alcoholic Fermentation Using HS-SPME and Liquid Injection with TMS Derivatization GC-MS Methods.Molecules. 2023 Jun 8;28(12):4653. doi: 10.3390/molecules28124653. Molecules. 2023. PMID: 37375206 Free PMC article.
-
Discovering the Influence of Microorganisms on Wine Color.Front Microbiol. 2021 Dec 3;12:790935. doi: 10.3389/fmicb.2021.790935. eCollection 2021. Front Microbiol. 2021. PMID: 34925298 Free PMC article. Review.
References
-
- Bartowsky E.J. Oenococcus oeni and malolactic fermentation—Moving into the molecular arena. Aust. J. Grape Wine Res. 2005;11:174–187. doi: 10.1111/j.1755-0238.2005.tb00286.x. - DOI
-
- Lerm E., Engelbrecht L., Du Toit M. Malolactic Fermentation: The ABC’s of MLF. S. Afr. J. Enol. Vitic. 2016;31:186–212. doi: 10.21548/31-2-1417. - DOI
-
- Davis C.R., Wibowo D., Eschenbruch R., Lee T.H., Fleet G.H. Practical Implications of Malolactic Fermentation: A Review. Am. J. Enol. Vitic. 1985;36:290–301.
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

