Inhibition of lysosomal phospholipase A2 predicts drug-induced phospholipidosis
- PMID: 34087196
- PMCID: PMC8243516
- DOI: 10.1016/j.jlr.2021.100089
Inhibition of lysosomal phospholipase A2 predicts drug-induced phospholipidosis
Abstract
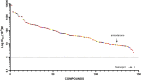
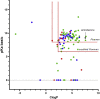
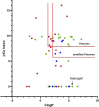
Phospholipidosis, the excessive accumulation of phospholipids within lysosomes, is a pathological response observed following exposure to many drugs across multiple therapeutic groups. A clear mechanistic understanding of the causes and implications of this form of drug toxicity has remained elusive. We previously reported the discovery and characterization of a lysosome-specific phospholipase A2 (PLA2G15) and later reported that amiodarone, a known cause of drug-induced phospholipidosis, inhibits this enzyme. Here, we assayed a library of 163 drugs for inhibition of PLA2G15 to determine whether this phospholipase was the cellular target for therapeutics other than amiodarone that cause phospholipidosis. We observed that 144 compounds inhibited PLA2G15 activity. Thirty-six compounds not previously reported to cause phospholipidosis inhibited PLA2G15 with IC50 values less than 1 mM and were confirmed to cause phospholipidosis in an in vitro assay. Within this group, fosinopril was the most potent inhibitor (IC50 0.18 μM). Additional characterization of the inhibition of PLA2G15 by fosinopril was consistent with interference of PLA2G15 binding to liposomes. PLA2G15 inhibition was more accurate in predicting phospholipidosis compared with in silico models based on pKa and ClogP, measures of protonation, and transport-independent distribution in the lysosome, respectively. In summary, PLA2G15 is a primary target for cationic amphiphilic drugs that cause phospholipidosis, and PLA2G15 inhibition by cationic amphiphilic compounds provides a potentially robust screening platform for potential toxicity during drug development.
Keywords: 1-O-acylceramide; Acyltransferase; amiodarone; cationic amphiphilic drugs; drug development; drug toxicity; drug-induced phospholipidosis; high-throughput screening; lysosome; phospholipase A2 group XV.
Published by Elsevier Inc.
Conflict of interest statement
Conflict of interest The authors declare that they have no conflicts of interest with the contents of this article. Recombinant LPLA(2) and anti-LPLA(2) monoclonal antibodies are licensed to Echelon Biosciences by the University of Michigan.
Figures

Similar articles
-
The role of lysosomal phospholipase A2 in the catabolism of bis(monoacylglycerol)phosphate and association with phospholipidosis.J Lipid Res. 2024 Jul;65(7):100574. doi: 10.1016/j.jlr.2024.100574. Epub 2024 Jun 9. J Lipid Res. 2024. PMID: 38857781 Free PMC article.
-
Lysosomal phospholipase A2.Biochim Biophys Acta Mol Cell Biol Lipids. 2019 Jun;1864(6):932-940. doi: 10.1016/j.bbalip.2018.07.012. Epub 2018 Aug 2. Biochim Biophys Acta Mol Cell Biol Lipids. 2019. PMID: 30077006 Free PMC article. Review.
-
A role for lysosomal phospholipase A2 in drug induced phospholipidosis.Drug Metab Lett. 2007 Jan;1(1):49-53. doi: 10.2174/187231207779814292. Drug Metab Lett. 2007. PMID: 19356018 Review.
-
Group XV phospholipase A₂, a lysosomal phospholipase A₂.Prog Lipid Res. 2011 Jan;50(1):1-13. doi: 10.1016/j.plipres.2010.10.006. Epub 2010 Nov 11. Prog Lipid Res. 2011. PMID: 21074554 Free PMC article. Review.
-
A toxicogenomic approach to drug-induced phospholipidosis: analysis of its induction mechanism and establishment of a novel in vitro screening system.Toxicol Sci. 2005 Feb;83(2):282-92. doi: 10.1093/toxsci/kfh264. Epub 2004 Sep 1. Toxicol Sci. 2005. PMID: 15342952
Cited by
-
Chemogenomics for NR1 nuclear hormone receptors.Nat Commun. 2024 Jun 18;15(1):5201. doi: 10.1038/s41467-024-49493-6. Nat Commun. 2024. PMID: 38890295 Free PMC article.
-
Lyciumbarbarum polysaccharides ameliorate canine acute liver injury by reducing oxidative stress, protecting mitochondrial function, and regulating metabolic pathways.J Zhejiang Univ Sci B. 2023 Feb 15;24(2):157-171. doi: 10.1631/jzus.B2200213. J Zhejiang Univ Sci B. 2023. PMID: 36751701 Free PMC article.
-
Glycerophosphodiesters inhibit lysosomal phospholipid catabolism in Batten disease.Mol Cell. 2024 Apr 4;84(7):1354-1364.e9. doi: 10.1016/j.molcel.2024.02.006. Epub 2024 Mar 5. Mol Cell. 2024. PMID: 38447580
-
The role of lysosomal phospholipase A2 in the catabolism of bis(monoacylglycerol)phosphate and association with phospholipidosis.J Lipid Res. 2024 Jul;65(7):100574. doi: 10.1016/j.jlr.2024.100574. Epub 2024 Jun 9. J Lipid Res. 2024. PMID: 38857781 Free PMC article.
-
The relevance of acid sphingomyelinase as a potential target for therapeutic intervention in hepatic disorders: current scenario and anticipated trends.Arch Toxicol. 2023 Aug;97(8):2069-2087. doi: 10.1007/s00204-023-03529-w. Epub 2023 May 29. Arch Toxicol. 2023. PMID: 37248308 Free PMC article. Review.
References
-
- Anderson N., Borlak J. Drug-induced phospholipidosis. FEBS Lett. 2006;580:5533–5540. - PubMed
-
- Chen G.L., Sutrina S.L., Frayer K.L., Chen W.W. Effects of lysosomotropic agents on lipogenesis. Arch. Biochem. Biophys. 1986;245:66–75. - PubMed
-
- Kubo M., Hostetler K.Y. Mechanism of cationic amphiphilic drug inhibition of purified lysosomal phospholipase A1. Biochemistry. 1985;24:6515–6520. - PubMed
-
- Ikeda K., Hirayama M., Hirota Y., Asa E., Seki J., Tanaka Y. Drug-induced phospholipidosis is caused by blockade of mannose 6-phosphate receptor-mediated targeting of lysosomal enzymes. Biochem. Biophys. Res. Commun. 2008;377:268–274. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

