Addressing personal protective equipment (PPE) decontamination: Methylene blue and light inactivates severe acute respiratory coronavirus virus 2 (SARS-CoV-2) on N95 respirators and medical masks with maintenance of integrity and fit
- PMID: 34016200
- PMCID: PMC8220024
- DOI: 10.1017/ice.2021.230
Addressing personal protective equipment (PPE) decontamination: Methylene blue and light inactivates severe acute respiratory coronavirus virus 2 (SARS-CoV-2) on N95 respirators and medical masks with maintenance of integrity and fit
Abstract
Objective: The coronavirus disease 2019 (COVID-19) pandemic has resulted in shortages of personal protective equipment (PPE), underscoring the urgent need for simple, efficient, and inexpensive methods to decontaminate masks and respirators exposed to severe acute respiratory coronavirus virus 2 (SARS-CoV-2). We hypothesized that methylene blue (MB) photochemical treatment, which has various clinical applications, could decontaminate PPE contaminated with coronavirus.
Design: The 2 arms of the study included (1) PPE inoculation with coronaviruses followed by MB with light (MBL) decontamination treatment and (2) PPE treatment with MBL for 5 cycles of decontamination to determine maintenance of PPE performance.
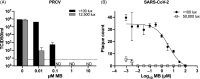
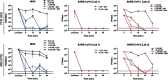
Methods: MBL treatment was used to inactivate coronaviruses on 3 N95 filtering facepiece respirator (FFR) and 2 medical mask models. We inoculated FFR and medical mask materials with 3 coronaviruses, including SARS-CoV-2, and we treated them with 10 µM MB and exposed them to 50,000 lux of white light or 12,500 lux of red light for 30 minutes. In parallel, integrity was assessed after 5 cycles of decontamination using multiple US and international test methods, and the process was compared with the FDA-authorized vaporized hydrogen peroxide plus ozone (VHP+O3) decontamination method.
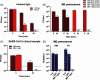
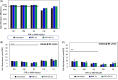
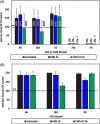
Results: Overall, MBL robustly and consistently inactivated all 3 coronaviruses with 99.8% to >99.9% virus inactivation across all FFRs and medical masks tested. FFR and medical mask integrity was maintained after 5 cycles of MBL treatment, whereas 1 FFR model failed after 5 cycles of VHP+O3.
Conclusions: MBL treatment decontaminated respirators and masks by inactivating 3 tested coronaviruses without compromising integrity through 5 cycles of decontamination. MBL decontamination is effective, is low cost, and does not require specialized equipment, making it applicable in low- to high-resource settings.
Figures
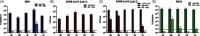
Similar articles
-
"Don, doff, discard" to "don, doff, decontaminate"-FFR and mask integrity and inactivation of a SARS-CoV-2 surrogate and a norovirus following multiple vaporised hydrogen peroxide-, ultraviolet germicidal irradiation-, and dry heat decontaminations.PLoS One. 2021 May 19;16(5):e0251872. doi: 10.1371/journal.pone.0251872. eCollection 2021. PLoS One. 2021. PMID: 34010337 Free PMC article.
-
Aerosolized Hydrogen Peroxide Decontamination of N95 Respirators, with Fit-Testing and Viral Inactivation, Demonstrates Feasibility for Reuse during the COVID-19 Pandemic.mSphere. 2022 Oct 26;7(5):e0030322. doi: 10.1128/msphere.00303-22. Epub 2022 Aug 30. mSphere. 2022. PMID: 36040048 Free PMC article.
-
Methylene blue applied to N95 respirators and medical masks for SARS-CoV-2 decontamination: What is the likelihood of inhaling methylene blue?Am J Infect Control. 2022 Aug;50(8):857-862. doi: 10.1016/j.ajic.2022.03.003. Am J Infect Control. 2022. PMID: 35908823 Free PMC article.
-
Principles and practice for SARS-CoV-2 decontamination of N95 masks with UV-C.Biophys J. 2021 Jul 20;120(14):2927-2942. doi: 10.1016/j.bpj.2021.02.039. Epub 2021 Mar 4. Biophys J. 2021. PMID: 33675766 Free PMC article. Review.
-
Rapid Review of SARS-CoV-1 and SARS-CoV-2 Viability, Susceptibility to Treatment, and the Disinfection and Reuse of PPE, Particularly Filtering Facepiece Respirators.Int J Environ Res Public Health. 2020 Aug 22;17(17):6117. doi: 10.3390/ijerph17176117. Int J Environ Res Public Health. 2020. PMID: 32842655 Free PMC article. Review.
Cited by
-
Exploring inactivation of SARS-CoV-2, MERS-CoV, Ebola, Lassa, and Nipah viruses on N95 and KN95 respirator material using photoactivated methylene blue to enable reuse.Am J Infect Control. 2022 Aug;50(8):863-870. doi: 10.1016/j.ajic.2022.02.016. Am J Infect Control. 2022. PMID: 35908824 Free PMC article.
-
Effect of five decontamination methods on face masks and filtering facepiece respirators contaminated with Staphylococcus aureus and Pseudomonas aeruginosa.Access Microbiol. 2022 Mar 30;4(3):000342. doi: 10.1099/acmi.0.000342. eCollection 2022. Access Microbiol. 2022. PMID: 35693470 Free PMC article.
-
Inactivation strategies for SARS-CoV-2 on surgical masks using light-activated chemical dyes.Am J Infect Control. 2022 Aug;50(8):844-848. doi: 10.1016/j.ajic.2022.03.015. Am J Infect Control. 2022. PMID: 35908821 Free PMC article.
-
Methylene blue in combination with sunlight as a low cost and effective disinfection method for coronavirus-contaminated PPE.Am J Infect Control. 2022 Aug;50(8):906-908. doi: 10.1016/j.ajic.2022.02.019. Am J Infect Control. 2022. PMID: 35908830 Free PMC article.
-
Filtration performance, fit test and side effects of respiratory personal protective equipment following decontamination: Observations for user safety and comfort.PLoS One. 2023 Jan 23;18(1):e0280426. doi: 10.1371/journal.pone.0280426. eCollection 2023. PLoS One. 2023. PMID: 36689487 Free PMC article.
References
-
- Rational use of personal protective equipment for coronavirus disease (COVID-19) and considerations during severe shortages. World Health Organization website. https://www.who.int/publications/i/item/rational-use-of-personal-protect.... Published December 23, 2020. Accessed May 3, 2021.
-
- Decontamination systems for personal protective equipment EUAs, 2020; 1–4. US Food and Drug Administration website. https://www.fda.gov/medical-devices/coronavirus-disease-2019-covid-19-em.... Accessed December 20, 2020.
-
- Richter W, Hofacre K, Willenberg Z. Final Report for the Bioquell hydrogen peroxide vapor (HPV) decontamination for reuse of N95 respirators, Battelle. US Food and Drug Administration website. https://www.fda.gov/media/136386/download. Published 2016. Accessed April 24, 2021.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

