Genetic mosaicism, intrafamilial phenotypic heterogeneity, and molecular defects of a novel missense SLC6A1 mutation associated with epilepsy and ADHD
- PMID: 33961861
- PMCID: PMC9116449
- DOI: 10.1016/j.expneurol.2021.113723
Genetic mosaicism, intrafamilial phenotypic heterogeneity, and molecular defects of a novel missense SLC6A1 mutation associated with epilepsy and ADHD
Abstract
Background: Mutations in SLC6A1, encoding γ-aminobutyric acid (GABA) transporter 1 (GAT-1), have been recently associated with a spectrum of neurodevelopmental disorders ranging from variable epilepsy syndromes, intellectual disability (ID), autism and others. To date, most identified mutations are de novo. We here report a pedigree of two siblings associated with myoclonic astatic epilepsy, attention deficit hyperactivity disorder (ADHD), and ID.
Methods: Next-generation sequencing identified a missense mutation in the SLC6A1 gene (c.373G > A(p.Val125Met)) in the sisters but not in their shared mother who is also asymptomatic, suggesting gonadal mosaicism. We have thoroughly characterized the clinical phenotypes: EEG recordings identified features for absence seizures and prominent bursts of occipital intermittent rhythmic delta activity (OIRDA). The molecular pathophysiology underlying the clinical phenotypes was assessed using a multidisciplinary approach including machine learning, confocal microscopy, and high-throughput 3H radio-labeled GABA uptake assays in mouse astrocytes and neurons.
Results: The GAT-1(Val125Met) mutation destabilizes the global protein conformation and reduces transporter protein expression at total and cell surface. The mutant transporter protein was localized intracellularly inside the endoplasmic reticulum (ER) in both HEK293T cells and astrocytes which may directly contribute to seizures in patients. Radioactive 3H-labeled GABA uptake assay indicated the mutation reduced the function of the mutant GAT-1(Val125Met) to ~30% of the wildtype.
Conclusions: The seizure phenotypes, ADHD, and impaired cognition are likely caused by a partial loss-of-function of GAT-1 due to protein destabilization resulting from the mutation. Reduced GAT-1 function in astrocytes and neurons may consequently alter brain network activities such as increased seizures and reduced attention.
Keywords: ADHD; GABA transporter 1; Mosaicism; Myoclonic atonic epilepsy (MAE); Phenotypic heterogeneity; Protein stability.
Copyright © 2021. Published by Elsevier Inc.
Conflict of interest statement
Competing interests
The authors declare that they are no competing interests.
Figures
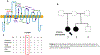


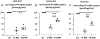
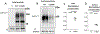

Similar articles
-
Astrocytic GABA transporter 1 deficit in novel SLC6A1 variants mediated epilepsy: Connected from protein destabilization to seizures in mice and humans.Neurobiol Dis. 2022 Oct 1;172:105810. doi: 10.1016/j.nbd.2022.105810. Epub 2022 Jul 14. Neurobiol Dis. 2022. PMID: 35840120 Free PMC article.
-
Endoplasmic reticulum retention and degradation of a mutation in SLC6A1 associated with epilepsy and autism.Mol Brain. 2020 May 12;13(1):76. doi: 10.1186/s13041-020-00612-6. Mol Brain. 2020. PMID: 32398021 Free PMC article.
-
Common molecular mechanisms of SLC6A1 variant-mediated neurodevelopmental disorders in astrocytes and neurons.Brain. 2021 Sep 4;144(8):2499-2512. doi: 10.1093/brain/awab207. Brain. 2021. PMID: 34028503 Free PMC article.
-
Molecular and Clinical Repercussions of GABA Transporter 1 Variants Gone Amiss: Links to Epilepsy and Developmental Spectrum Disorders.Front Mol Biosci. 2022 Mar 2;9:834498. doi: 10.3389/fmolb.2022.834498. eCollection 2022. Front Mol Biosci. 2022. PMID: 35295842 Free PMC article. Review.
-
MED13 mutation: A novel cause of developmental and epileptic encephalopathy with infantile spasms.Seizure. 2022 Oct;101:211-217. doi: 10.1016/j.seizure.2022.09.002. Epub 2022 Sep 3. Seizure. 2022. PMID: 36087421 Review.
Cited by
-
Intrafamilial variability in SLC6A1-related neurodevelopmental disorders.Front Neurosci. 2023 Jul 12;17:1219262. doi: 10.3389/fnins.2023.1219262. eCollection 2023. Front Neurosci. 2023. PMID: 37502687 Free PMC article.
-
Astrocytic GABA transporter 1 deficit in novel SLC6A1 variants mediated epilepsy: Connected from protein destabilization to seizures in mice and humans.Neurobiol Dis. 2022 Oct 1;172:105810. doi: 10.1016/j.nbd.2022.105810. Epub 2022 Jul 14. Neurobiol Dis. 2022. PMID: 35840120 Free PMC article.
-
SLC6A1 variant pathogenicity, molecular function and phenotype: a genetic and clinical analysis.Brain. 2023 Dec 1;146(12):5198-5208. doi: 10.1093/brain/awad292. Brain. 2023. PMID: 37647852 Free PMC article.
-
γ-Aminobutyric acid transporter and GABAA receptor mechanisms in Slc6a1+/A288V and Slc6a1+/S295L mice associated with developmental and epileptic encephalopathies.Brain Commun. 2024 Apr 22;6(2):fcae110. doi: 10.1093/braincomms/fcae110. eCollection 2024. Brain Commun. 2024. PMID: 38650830 Free PMC article.
-
A transporter's doom or destiny: SLC6A1 in health and disease, novel molecular targets and emerging therapeutic prospects.Front Mol Neurosci. 2024 Aug 29;17:1466694. doi: 10.3389/fnmol.2024.1466694. eCollection 2024. Front Mol Neurosci. 2024. PMID: 39268250 Free PMC article. Review.
References
-
- Carvill GL, McMahon JM, Schneider A, Zemel M, Myers CT, Saykally J, Nguyen J, Robbiano A, Zara F, Specchio N, Mecarelli O, Smith RL, Leventer RJ, Moller RS, Nikanorova M, Dimova P, Jordanova A, Petrou S, Helbig I, Striano P, Weckhuysen S, Berkovic SF, Scheffer IE, Mefford HC. Mutations in the GABA Transporter SLC6A1 Cause Epilepsy with Myoclonic-Atonic Seizures. Am J Hum Genet 2015. May 7;96(5):808–15. - PMC - PubMed
-
- Johannesen KM, Gardella E, Linnankivi T, Courage C, de Saint MA, Lehesjoki AE, Mignot C, Afenjar A, Lesca G, Abi-Warde MT, Chelly J, Piton A, Merritt JL, Rodan LH, Tan WH, Bird LM, Nespeca M, Gleeson JG, Yoo Y, Choi M, Chae JH, Czapansky-Beilman D, Reichert SC, Pendziwiat M, Verhoeven JS, Schelhaas HJ, Devinsky O, Christensen J, Specchio N, Trivisano M, Weber YG, Nava C, Keren B, Doummar D, Schaefer E, Hopkins S, Dubbs H, Shaw JE, Pisani L, Myers CT, Tang S, Tang S, Pal DK, Millichap JJ, Carvill GL, Helbig KL, Mecarelli O, Striano P, Helbig I, Rubboli G, Mefford HC, Moller RS. Defining the phenotypic spectrum of SLC6A1 mutations. Epilepsia 2018. Feb;59(2):389–402. - PMC - PubMed
-
- Mattison KA, Butler KM, Inglis GAS, Dayan O, Boussidan H, Bhambhani V, Philbrook B, da SC, Alexander JJ, Kanner BI, Escayg A. SLC6A1 variants identified in epilepsy patients reduce gamma-aminobutyric acid transport. Epilepsia 2018. Sep;59(9):e135–e141. - PubMed
-
- Cai K, Wang J, Eissman J, Wang J, Nwosu G, Shen W, Liang HC, Li XJ, Zhu HX, Yi YH, Song J, Xu D, Delpire E, Liao WP, Shi YW, Kang JQ. A missense mutation in SLC6A1 associated with Lennox-Gastaut syndrome impairs GABA transporter 1 protein trafficking and function. Exp Neurol 2019. Jun 6;320:112973. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

