Donanemab (LY3002813) dose-escalation study in Alzheimer's disease
- PMID: 33614890
- PMCID: PMC7882532
- DOI: 10.1002/trc2.12112
Donanemab (LY3002813) dose-escalation study in Alzheimer's disease
Abstract
Introduction: This study explored the safety and tolerability features of donanemab (LY3002813) in patients with mild cognitive impairment due to Alzheimer's disease (AD) or mild to moderate AD dementia.
Methods: Patients with AD were enrolled into the single-ascending dose phase and were administered a single, intravenous (IV) dose of donanemab (five dosing cohorts from 0.1 to 10 mg/kg) or placebo followed by a 12-week follow-up period for each dose level. After the follow-up period, the same patients proceeded into the multiple-ascending dose (MAD) phase (five cohorts) and were administered IV doses of donanemab (0.3 to 10 mg/kg) or placebo approximately once per month for up to four doses depending on the initial doses (only cohort 1 went from 0.1 mg/kg to a higher dose of 0.3 mg/kg during the MAD phase). This phase concluded with a 12-week follow-up period. The relative exposure assessment of an unblinded, single, subcutaneous 3-mg/kg dose of donanemab in patients with AD was also performed, followed by a 12-week follow-up period. One cohort of healthy subjects received an unblinded, single, IV 1-mg/kg dose of donanemab. These two cohorts did not continue to the MAD phase.
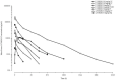
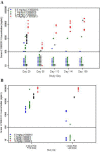
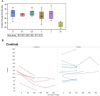
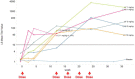
Results: Donanemab was generally well tolerated up to 10 mg/kg. After single-dose administration from 0.1 to 3.0 mg/kg, the mean terminal elimination half-life was ≈4 days, increasing to ≈10 days at 10 mg/kg. Only the 10-mg/kg dose showed changes in amyloid positron emission tomography. Amyloid reduction of 40% to 50% was achieved. Approximately 90% of subjects developed anti-drug antibodies at 3 months after a single intravenous dose.
Discussion: Intravenous donanemab 10 mg/kg can reduce amyloid deposits in AD despite having a shorter than expected half-life.
Keywords: Alzheimer's disease; dementia; donanemab; mild to moderate; tolerability.
© 2020 The Authors. Alzheimer's & Dementia: Translational Research & Clinical Interventions published by Wiley Periodicals, Inc. on behalf of Alzheimer's Association.
Conflict of interest statement
Authors are employees and minor stockholders of Eli Lilly and Company. Anne Hawdon is a former employee of Eli Lilly and Company.
Figures

Similar articles
-
Donanemab (LY3002813) Phase 1b Study in Alzheimer's Disease: Rapid and Sustained Reduction of Brain Amyloid Measured by Florbetapir F18 Imaging.J Prev Alzheimers Dis. 2021;8(4):414-424. doi: 10.14283/jpad.2021.56. J Prev Alzheimers Dis. 2021. PMID: 34585215 Clinical Trial.
-
Association of Amyloid Reduction After Donanemab Treatment With Tau Pathology and Clinical Outcomes: The TRAILBLAZER-ALZ Randomized Clinical Trial.JAMA Neurol. 2022 Oct 1;79(10):1015-1024. doi: 10.1001/jamaneurol.2022.2793. JAMA Neurol. 2022. PMID: 36094645 Free PMC article. Clinical Trial.
-
Pharmacokinetic evaluation of donanemab for the treatment of Alzheimer's.Expert Opin Drug Metab Toxicol. 2024 Jun;20(6):411-417. doi: 10.1080/17425255.2024.2357637. Epub 2024 May 20. Expert Opin Drug Metab Toxicol. 2024. PMID: 38758223 Review.
-
Donanemab Population Pharmacokinetics, Amyloid Plaque Reduction, and Safety in Participants with Alzheimer's Disease.Clin Pharmacol Ther. 2023 Jun;113(6):1258-1267. doi: 10.1002/cpt.2875. Epub 2023 Mar 9. Clin Pharmacol Ther. 2023. PMID: 36805552 Clinical Trial.
-
How donanemab data address the coverage with evidence development questions.Alzheimers Dement. 2024 Apr;20(4):3127-3140. doi: 10.1002/alz.13700. Epub 2024 Feb 7. Alzheimers Dement. 2024. PMID: 38323738 Free PMC article. Review.
Cited by
-
New Insights into Alzheimer's Disease: Novel Pathogenesis, Drug Target and Delivery.Pharmaceutics. 2023 Apr 3;15(4):1133. doi: 10.3390/pharmaceutics15041133. Pharmaceutics. 2023. PMID: 37111618 Free PMC article. Review.
-
Emerging Alzheimer's disease therapeutics: promising insights from lipid metabolism and microglia-focused interventions.Front Aging Neurosci. 2023 Oct 25;15:1259012. doi: 10.3389/fnagi.2023.1259012. eCollection 2023. Front Aging Neurosci. 2023. PMID: 38020773 Free PMC article. Review.
-
Promising candidates from drug clinical trials: Implications for clinical treatment of Alzheimer's disease in China.Front Neurol. 2022 Nov 15;13:1034243. doi: 10.3389/fneur.2022.1034243. eCollection 2022. Front Neurol. 2022. PMID: 36457865 Free PMC article. Review.
-
Disease-Modifying Therapies for Alzheimer's Disease: More Questions than Answers.Neurotherapeutics. 2022 Jan;19(1):209-227. doi: 10.1007/s13311-022-01201-2. Epub 2022 Feb 28. Neurotherapeutics. 2022. PMID: 35229269 Free PMC article. Review.
-
Targeting alpha-synuclein via the immune system in Parkinson's disease: Current vaccine therapies.Neuropharmacology. 2022 Jan 1;202:108870. doi: 10.1016/j.neuropharm.2021.108870. Epub 2021 Nov 3. Neuropharmacology. 2022. PMID: 34742741 Free PMC article. Review.
References
-
- DeMattos RB, Lu J, Tang Y, et al. A plaque‐specifc antibody clears existing β‐amyloid plaques in Alzheimer's disease mice. Neuron 2012;76(5):908‐920. - PubMed
-
- Barthel H, Gertz H‐J, Dresel S, et al. Florbetaben Study Group . Cerebral amyloid‐β PET with florbetaben (18F) in patients with Alzheimer's disease and healthy controls: a multicentre phase 2 diagnostic study. Lancet Neurol 2011;10(5):424‐435. - PubMed
-
- Navitsky M, Joshi AD, Kennedy I, et al. Standardization of amyloid quantitation with florbetapir standardized uptake value ratios to the Centiloid scale. Alzheimers Dement 2018;14(12):1565‐1571. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
