Atogepant Is Not Associated With Clinically Meaningful Alanine Aminotransferase Elevations in Healthy Adults
- PMID: 33142014
- PMCID: PMC7993278
- DOI: 10.1111/cts.12917
Atogepant Is Not Associated With Clinically Meaningful Alanine Aminotransferase Elevations in Healthy Adults
Abstract
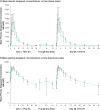
Atogepant is a potent, selective, oral calcitonin gene-related peptide (CGRP) receptor antagonist in development for migraine prevention. The chemical structure of atogepant is distinct from previous CGRP receptor antagonists, which were associated with elevated serum alanine aminotransferase (ALT) in clinical trials. Here, we report the safety, tolerability, and pharmacokinetics (PKs) of a once-daily supratherapeutic dose (170 mg) of atogepant for 28 days from a randomized, double-blind, placebo-controlled phase I trial in healthy participants. Overall safety, hepatic safety, and plasma PK parameters were evaluated. Thirty-four participants aged 23-55 years enrolled; 28 (82.4%) completed the study in accordance with the protocol. Multiple doses of 170 mg atogepant for 28 consecutive days were generally well-tolerated. All adverse events (AEs; reported in 87.0% of the atogepant group; 72.7%, placebo) were mild in severity except one serious AE of subarachnoid hemorrhage due to a bicycle accident and not considered related to treatment. There were two discontinuations due to AEs, both with atogepant, one considered possibly related to treatment. Over 28 days of treatment, no participant receiving atogepant had an ALT elevation above 1.5 × upper limit of normal. Change from baseline in serum ALT levels was not different between atogepant and placebo. Atogepant is rapidly absorbed (median time to maximum plasma concentration, ~ 2 hours) with an apparent terminal half-life of ~ 11 hours, and no evidence of accumulation after once-daily dosing. Overall, atogepant at a high oral dose is safe and well-tolerated in healthy participants with no clinically meaningful elevations in ALT.
Trial registration: ClinicalTrials.gov NCT03700320 NCT03777059 NCT03855137 NCT03939312.
© 2020 Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc. Clinical and Translational Science published by Wiley Periodicals LLC on behalf of the American Society for Clinical Pharmacology and Therapeutics.
Conflict of interest statement
K.C.M., J.X., and L.M. were employees of Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA, with stock in Merck & Co., Inc., Kenilworth, NJ, USA, at the time of this study. W.K.K. served as a consultant to Merck in 2019 for a topic unrelated to atogepant. P.B., F.C.‐G., W.L., D.P., M.F.D., and C.Z.M. are employees of Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA, with stock in Merck & Co., Inc., Kenilworth, NJ, USA. R.B. is an employee of AbbVie, and may hold AbbVie stock.
Figures

Similar articles
-
A Single Supratherapeutic Dose of Atogepant Does Not Affect Cardiac Repolarization in Healthy Adults: Results From a Randomized, Single-Dose, Phase 1 Crossover Trial.Clin Pharmacol Drug Dev. 2021 Sep;10(9):1099-1107. doi: 10.1002/cpdd.940. Epub 2021 May 4. Clin Pharmacol Drug Dev. 2021. PMID: 33942560 Free PMC article. Clinical Trial.
-
Single-Dose Pharmacokinetics and Safety of Atogepant in Adults With Hepatic Impairment: Results From an Open-Label, Phase 1 Trial.Clin Pharmacol Drug Dev. 2021 Jul;10(7):726-733. doi: 10.1002/cpdd.916. Epub 2021 Jan 27. Clin Pharmacol Drug Dev. 2021. PMID: 33501783 Free PMC article. Clinical Trial.
-
Atogepant for the Preventive Treatment of Migraine.N Engl J Med. 2021 Aug 19;385(8):695-706. doi: 10.1056/NEJMoa2035908. N Engl J Med. 2021. PMID: 34407343 Clinical Trial.
-
Atogepant: First Approval.Drugs. 2022 Jan;82(1):65-70. doi: 10.1007/s40265-021-01644-5. Drugs. 2022. PMID: 34813050 Review.
-
Comparative efficacy, quality of life, safety, and tolerability of atogepant and rimegepant in migraine prevention: A matching-adjusted indirect comparison analysis.Cephalalgia. 2024 Feb;44(2):3331024241235156. doi: 10.1177/03331024241235156. Cephalalgia. 2024. PMID: 38410850 Review.
Cited by
-
Novel Oral CGRP Receptor Antagonist Atogepant in the Prevention of Migraine.Discoveries (Craiova). 2023 Jun 30;11(2):e167. doi: 10.15190/d.2023.6. eCollection 2023 Apr-Jun. Discoveries (Craiova). 2023. PMID: 37564925 Free PMC article. Review.
-
A Brief Review of Gepants.Curr Pain Headache Rep. 2023 Sep;27(9):479-488. doi: 10.1007/s11916-023-01142-1. Epub 2023 Aug 2. Curr Pain Headache Rep. 2023. PMID: 37531032 Review.
-
Migraine: from pathophysiology to treatment.J Neurol. 2023 Jul;270(7):3654-3666. doi: 10.1007/s00415-023-11706-1. Epub 2023 Apr 8. J Neurol. 2023. PMID: 37029836 Free PMC article. Review.
-
Gepants - a long way to cure: a narrative review.Neurol Sci. 2022 Sep;43(9):5697-5708. doi: 10.1007/s10072-022-06184-8. Epub 2022 Jun 2. Neurol Sci. 2022. PMID: 35650458 Free PMC article. Review.
-
Calcitonin Gene-Related Peptide (CGRP)-Targeted Monoclonal Antibodies and Antagonists in Migraine: Current Evidence and Rationale.BioDrugs. 2022 May;36(3):341-358. doi: 10.1007/s40259-022-00530-0. Epub 2022 Apr 27. BioDrugs. 2022. PMID: 35476215 Free PMC article. Review.
References
-
- Pietrobon, D. & Moskowitz, M.A. Pathophysiology of migraine. Annu. Rev. Physiol. 75, 365–391 (2013). - PubMed
-
- Burch, R.C. , Buse, D.C. & Lipton, R.B. Migraine: epidemiology, burden, and comorbidity. Neurol. Clin. 37, 631–649 (2019). - PubMed
-
- GBD 2016 Disease and Injury Incidence and Prevalence Collaborators . Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet 390, 1211–1259 (2017). - PMC - PubMed
-
- Lipton, R.B. , Bigal, M.E. , Diamond, M. , Freitag, F. , Reed, M.L. & Stewart, W.F. Migraine prevalence, disease burden, and the need for preventive therapy. Neurology 68, 343–349 (2007). - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

