Loss-of-Function Variants in PPP1R12A: From Isolated Sex Reversal to Holoprosencephaly Spectrum and Urogenital Malformations
- PMID: 31883643
- PMCID: PMC7042489
- DOI: 10.1016/j.ajhg.2019.12.004
Loss-of-Function Variants in PPP1R12A: From Isolated Sex Reversal to Holoprosencephaly Spectrum and Urogenital Malformations
Abstract
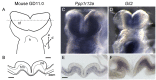
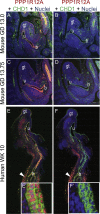
In two independent ongoing next-generation sequencing projects for individuals with holoprosencephaly and individuals with disorders of sex development, and through international research collaboration, we identified twelve individuals with de novo loss-of-function (LoF) variants in protein phosphatase 1, regulatory subunit 12a (PPP1R12A), an important developmental gene involved in cell migration, adhesion, and morphogenesis. This gene has not been previously reported in association with human disease, and it has intolerance to LoF as illustrated by a very low observed-to-expected ratio of LoF variants in gnomAD. Of the twelve individuals, midline brain malformations were found in five, urogenital anomalies in nine, and a combination of both phenotypes in two. Other congenital anomalies identified included omphalocele, jejunal, and ileal atresia with aberrant mesenteric blood supply, and syndactyly. Six individuals had stop gain variants, five had a deletion or duplication resulting in a frameshift, and one had a canonical splice acceptor site loss. Murine and human in situ hybridization and immunostaining revealed PPP1R12A expression in the prosencephalic neural folds and protein localization in the lower urinary tract at critical periods for forebrain division and urogenital development. Based on these clinical and molecular findings, we propose the association of PPP1R12A pathogenic variants with a congenital malformations syndrome affecting the embryogenesis of the brain and genitourinary systems and including disorders of sex development.
Keywords: MYPT1; PPP1R12A; disorders of sex development; embryogenesis; encephalocele; facial dysmorphism; forebrain; holoprosencephaly; hypospadias; omphalocele.
Published by Elsevier Inc.
Conflict of interest statement
The authors declare no competing interests.
Figures
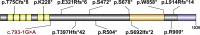
Similar articles
-
PPP1R12A Pathogenic Variants Associated with Human Congenital Malformations Syndrome.Am J Med Genet A. 2020 Apr;182(4):616-617. doi: 10.1002/ajmg.a.61525. Am J Med Genet A. 2020. PMID: 32153129 No abstract available.
-
A CCR4-NOT Transcription Complex, Subunit 1, CNOT1, Variant Associated with Holoprosencephaly.Am J Hum Genet. 2019 May 2;104(5):990-993. doi: 10.1016/j.ajhg.2019.03.017. Epub 2019 Apr 18. Am J Hum Genet. 2019. PMID: 31006510 Free PMC article.
-
Cerebral midline developmental anomalies: spectrum and associated features.Genet Couns. 1990;1(3-4):197-210. Genet Couns. 1990. PMID: 2098044 Review.
-
Cohesin complex-associated holoprosencephaly.Brain. 2019 Sep 1;142(9):2631-2643. doi: 10.1093/brain/awz210. Brain. 2019. PMID: 31334757 Free PMC article.
-
Holoprosencephaly: from Homer to Hedgehog.Clin Genet. 1998 Mar;53(3):155-63. doi: 10.1111/j.1399-0004.1998.tb02666.x. Clin Genet. 1998. PMID: 9630065 Review.
Cited by
-
Transposable element-derived sequences in vertebrate development.Mob DNA. 2021 Jan 6;12(1):1. doi: 10.1186/s13100-020-00229-5. Mob DNA. 2021. PMID: 33407840 Free PMC article. Review.
-
Heterozygous variants in SIX3 and POU1F1 cause pituitary hormone deficiency in mouse and man.Hum Mol Genet. 2023 Jan 13;32(3):367-385. doi: 10.1093/hmg/ddac192. Hum Mol Genet. 2023. PMID: 35951005 Free PMC article.
-
New SHH and Known SIX3 Variants in a Series of Latin American Patients with Holoprosencephaly.Mol Syndromol. 2021 Jul;12(4):219-233. doi: 10.1159/000515044. Epub 2021 Jun 15. Mol Syndromol. 2021. PMID: 34421500 Free PMC article.
-
Regulation of myosin light-chain phosphorylation and its roles in cardiovascular physiology and pathophysiology.Hypertens Res. 2022 Jan;45(1):40-52. doi: 10.1038/s41440-021-00733-y. Epub 2021 Oct 6. Hypertens Res. 2022. PMID: 34616031 Review.
-
Gene-environment interactions: aligning birth defects research with complex etiology.Development. 2020 Jul 17;147(21):dev191064. doi: 10.1242/dev.191064. Development. 2020. PMID: 32680836 Free PMC article. Review.
References
-
- Grassie M.E., Moffat L.D., Walsh M.P., MacDonald J.A. The myosin phosphatase targeting protein (MYPT) family: a regulated mechanism for achieving substrate specificity of the catalytic subunit of protein phosphatase type 1δ. Arch. Biochem. Biophys. 2011;510:147–159. - PubMed
-
- Kiss A., Erdődi F., Lontay B. Myosin phosphatase: Unexpected functions of a long-known enzyme. Biochim. Biophys. Acta Mol. Cell Res. 2019;1866:2–15. - PubMed
-
- Ito M., Nakano T., Erdodi F., Hartshorne D.J. Myosin phosphatase: structure, regulation and function. Mol. Cell. Biochem. 2004;259:197–209. - PubMed
-
- Shimizu H., Ito M., Miyahara M., Ichikawa K., Okubo S., Konishi T., Naka M., Tanaka T., Hirano K., Hartshorne D.J. Characterization of the myosin-binding subunit of smooth muscle myosin phosphatase. J. Biol. Chem. 1994;269:30407–30411. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

