Drug Screen Identifies Leflunomide for Treatment of Inflammatory Bowel Disease Caused by TTC7A Deficiency
- PMID: 31743734
- PMCID: PMC7062591
- DOI: 10.1053/j.gastro.2019.11.019
Drug Screen Identifies Leflunomide for Treatment of Inflammatory Bowel Disease Caused by TTC7A Deficiency
Abstract
Background & aims: Mutations in the tetratricopeptide repeat domain 7A gene (TTC7A) cause intestinal epithelial and immune defects. Patients can become immune deficient and develop apoptotic enterocolitis, multiple intestinal atresia, and recurrent intestinal stenosis. The intestinal disease in patients with TTC7A deficiency is severe and untreatable, and it recurs despite resection or allogeneic hematopoietic stem cell transplant. We screened drugs for those that prevent apoptosis of in cells with TTC7A deficiency and tested their effects in an animal model of the disease.
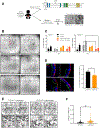
Methods: We developed a high-throughput screen to identify compounds approved by the US Food and Drug Administration that reduce activity of caspases 3 and 7 in TTC7A-knockout (TTC7A-KO) HAP1 (human haploid) cells and reduce the susceptibility to apoptosis. We validated the effects of identified agents in HeLa cells that stably express TTC7A with point mutations found in patients. Signaling pathways in cells were analyzed by immunoblots. We tested the effects of identified agents in zebrafish with disruption of ttc7a, which develop intestinal defects, and colonoids derived from biopsy samples of patients with and without mutations in TTC7A. We performed real-time imaging of intestinal peristalsis in zebrafish and histologic analyses of intestinal tissues from patients and zebrafish. Colonoids were analyzed by immunofluorescence and for ion transport.
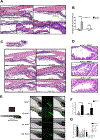
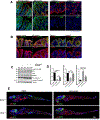
Results: TTC7A-KO HAP1 cells have abnormal morphology and undergo apoptosis, due to increased levels of active caspases 3 and 7. We identified drugs that increased cell viability; leflunomide (used to treat patients with inflammatory conditions) reduced caspase 3 and 7 activity in cells by 96%. TTC7A-KO cells contained cleaved caspase 3 and had reduced levels of phosphorylated AKT and X-linked inhibitor of apoptosis (XIAP); incubation of these cells with leflunomide increased levels of phosphorylated AKT and XIAP and reduced levels of cleaved caspase 3. Administration of leflunomide to ttc7a-/- zebrafish increased gut motility, reduced intestinal tract narrowing, increased intestinal cell survival, increased sizes of intestinal luminal spaces, and restored villi and goblet cell morphology. Exposure of patient-derived colonoids to leflunomide increased cell survival, polarity, and transport function.
Conclusions: In a drug screen, we identified leflunomide as an agent that reduces apoptosis and activates AKT signaling in TTC7A-KO cells. In zebrafish with disruption of ttc7a, leflunomide restores gut motility, reduces intestinal tract narrowing, and increases intestinal cell survival. This drug might be repurposed for treatment of TTC7A deficiency.
Keywords: Animal Model; Cell Death; Genetic; Monogenic IBD.
Copyright © 2020 AGA Institute. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Competing interests.
The authors declare no competing interests.
Figures


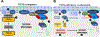
Similar articles
-
A Systematic Review of Monogenic Inflammatory Bowel Disease.Clin Gastroenterol Hepatol. 2022 Apr;20(4):e653-e663. doi: 10.1016/j.cgh.2021.03.021. Epub 2021 Mar 18. Clin Gastroenterol Hepatol. 2022. PMID: 33746097 Free PMC article. Review.
-
Mutations in tetratricopeptide repeat domain 7A result in a severe form of very early onset inflammatory bowel disease.Gastroenterology. 2014 Apr;146(4):1028-39. doi: 10.1053/j.gastro.2014.01.015. Epub 2014 Jan 11. Gastroenterology. 2014. PMID: 24417819 Free PMC article.
-
Immune deficiency-related enteropathy-lymphocytopenia-alopecia syndrome results from tetratricopeptide repeat domain 7A deficiency.J Allergy Clin Immunol. 2014 Dec;134(6):1354-1364.e6. doi: 10.1016/j.jaci.2014.07.019. Epub 2014 Aug 28. J Allergy Clin Immunol. 2014. PMID: 25174867
-
Whole-exome sequencing identifies tetratricopeptide repeat domain 7A (TTC7A) mutations for combined immunodeficiency with intestinal atresias.J Allergy Clin Immunol. 2013 Sep;132(3):656-664.e17. doi: 10.1016/j.jaci.2013.06.013. Epub 2013 Jul 4. J Allergy Clin Immunol. 2013. PMID: 23830146 Free PMC article.
-
Inhibitors of apoptosis (IAPs) regulate intestinal immunity and inflammatory bowel disease (IBD) inflammation.Trends Mol Med. 2014 Nov;20(11):652-65. doi: 10.1016/j.molmed.2014.09.006. Epub 2014 Oct 1. Trends Mol Med. 2014. PMID: 25282548 Review.
Cited by
-
Overcoming problematic growth phenotypes in organoids from patients with monogenic GI disease.PLoS One. 2024 Nov 4;19(11):e0309072. doi: 10.1371/journal.pone.0309072. eCollection 2024. PLoS One. 2024. PMID: 39495812 Free PMC article.
-
Congenital Diarrhoeas and Enteropathies.Nutrients. 2024 Sep 3;16(17):2971. doi: 10.3390/nu16172971. Nutrients. 2024. PMID: 39275287 Free PMC article. Review.
-
A Systematic Review of Monogenic Inflammatory Bowel Disease.Clin Gastroenterol Hepatol. 2022 Apr;20(4):e653-e663. doi: 10.1016/j.cgh.2021.03.021. Epub 2021 Mar 18. Clin Gastroenterol Hepatol. 2022. PMID: 33746097 Free PMC article. Review.
-
A synonymous mutation in PI4KA impacts the transcription and translation process of gene expression.Front Immunol. 2022 Oct 19;13:987666. doi: 10.3389/fimmu.2022.987666. eCollection 2022. Front Immunol. 2022. PMID: 36341355 Free PMC article.
-
Zebrafish Models of LAMA2-Related Congenital Muscular Dystrophy (MDC1A).Front Mol Neurosci. 2020 Jul 9;13:122. doi: 10.3389/fnmol.2020.00122. eCollection 2020. Front Mol Neurosci. 2020. PMID: 32742259 Free PMC article.
References
-
- Kammermeier J, Lucchini G, Pai SY, et al. Stem cell transplantation for tetratricopeptide repeat domain 7A deficiency: long-term follow-up. Blood 2016;128:1306–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

