Safety and convenience of once-weekly somapacitan in adult GH deficiency: a 26-week randomized, controlled trial
- PMID: 29500310
- PMCID: PMC5920019
- DOI: 10.1530/EJE-17-1073
Safety and convenience of once-weekly somapacitan in adult GH deficiency: a 26-week randomized, controlled trial
Abstract
Objective: Somapacitan is a reversible albumin-binding growth hormone (GH) derivative, developed for once-weekly administration. This study aimed to evaluate the safety of once-weekly somapacitan vs once-daily Norditropin®. Local tolerability and treatment satisfaction were also assessed.
Design: 26-week randomized, controlled phase 3 safety and tolerability trial in six countries (Nbib2382939).
Methods: Male or female patients aged 18-79 years with adult GH deficiency (AGHD), treated with once-daily GH for ≥6 months, were randomized to once-weekly somapacitan (n = 61) or once-daily Norditropin (n = 31) administered subcutaneously by pen. Both treatments were dose titrated for 8 weeks to achieve insulin-like growth factor I (IGF-I) standard deviation score (SDS) levels within the normal range, and then administered at a fixed dose. Outcome measures were adverse events (AEs), including injection site reactions; occurrence of anti-somapacitan/anti-GH antibodies and change in treatment satisfaction, assessed using the Treatment Satisfaction Questionnaire for Medication-9 (TSQM-9).
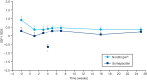
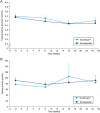
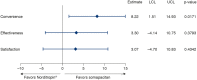
Results: Mean IGF-I SDS remained between 0 and 2 SDS throughout the trial in both groups. AEs were mostly mild or moderate and transient in nature. The most common AEs were nasopharyngitis, headache and fatigue in both groups. More than 1500 somapacitan injections were administered and no clinically significant injection site reactions were reported. No anti-somapacitan or anti-GH antibodies were detected. The TSQM-9 score for convenience increased significantly more with somapacitan vs Norditropin (P = 0.0171).
Conclusions: In this 26-week trial in patients with AGHD, somapacitan was well tolerated and no safety issues were identified. Once-weekly somapacitan was reported to be more convenient than once-daily Norditropin.
© 2018 The authors.
Figures
Similar articles
-
Somapacitan, a once-weekly reversible albumin-binding GH derivative, in children with GH deficiency: A randomized dose-escalation trial.Clin Endocrinol (Oxf). 2017 Oct;87(4):350-358. doi: 10.1111/cen.13409. Epub 2017 Aug 8. Clin Endocrinol (Oxf). 2017. PMID: 28656605 Clinical Trial.
-
Once-Weekly Somapacitan vs Daily GH in Children With GH Deficiency: Results From a Randomized Phase 2 Trial.J Clin Endocrinol Metab. 2020 Apr 1;105(4):e1847-61. doi: 10.1210/clinem/dgz310. J Clin Endocrinol Metab. 2020. PMID: 31917835 Free PMC article. Clinical Trial.
-
Once-weekly Somapacitan is Effective and Well Tolerated in Adults with GH Deficiency: A Randomized Phase 3 Trial.J Clin Endocrinol Metab. 2020 Apr 1;105(4):e1358-76. doi: 10.1210/clinem/dgaa049. J Clin Endocrinol Metab. 2020. PMID: 32022863 Free PMC article. Clinical Trial.
-
Utilizing Somapacitan, a Long-acting Growth Hormone Formulation, for the Treatment of Adult Growth Hormone Deficiency: A Guide for Clinicians.Endocr Pract. 2024 Oct;30(10):1003-1010. doi: 10.1016/j.eprac.2024.07.003. Epub 2024 Jul 9. Endocr Pract. 2024. PMID: 38992799 Review.
-
Once-Weekly Somapacitan as an Alternative Management of Growth Hormone Deficiency in Prepubertal Children: A Systematic Review and Meta-Analysis of Randomized Controlled Trial.Children (Basel). 2024 Feb 9;11(2):227. doi: 10.3390/children11020227. Children (Basel). 2024. PMID: 38397339 Free PMC article. Review.
Cited by
-
Dose-exposure-IGF-I response of once-weekly somapacitan in adults with GH deficiency.Eur J Endocrinol. 2022 May 16;187(1):27-38. doi: 10.1530/EJE-21-1167. Eur J Endocrinol. 2022. PMID: 35521713 Free PMC article. Clinical Trial.
-
Somapacitan-induced reversible lipoatrophy in an adult woman with hypopituitarism.Pituitary. 2024 Oct;27(5):737-739. doi: 10.1007/s11102-024-01440-w. Epub 2024 Aug 9. Pituitary. 2024. PMID: 39120809 Free PMC article.
-
Developments in the Management of Growth Hormone Deficiency: Clinical Utility of Somapacitan.Drug Des Devel Ther. 2024 Feb 3;18:291-306. doi: 10.2147/DDDT.S315172. eCollection 2024. Drug Des Devel Ther. 2024. PMID: 38333899 Free PMC article. Review.
-
Usefulness and Potential Pitfalls of Long-Acting Growth Hormone Analogs.Front Endocrinol (Lausanne). 2021 Feb 24;12:637209. doi: 10.3389/fendo.2021.637209. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 33716988 Free PMC article. Review.
-
Weekly somapacitan had no adverse effects on glucose metabolism in adults with growth hormone deficiency.Pituitary. 2023 Feb;26(1):57-72. doi: 10.1007/s11102-022-01283-3. Epub 2022 Nov 15. Pituitary. 2023. PMID: 36380045 Free PMC article.
References
-
- Carroll PV, Christ ER, Bengtsson BA, Carlsson L, Christiansen JS, Clemmons D, Hintz R, Ho K, Laron Z, Sizonenko P. et al Growth hormone deficiency in adulthood and the effects of growth hormone replacement: a review. Growth Hormone Research Society Scientific Committee. Journal of Clinical Endocrinology and Metabolism 1998. 83 382–395. (10.1210/jcem.83.2.4594) - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

