Epidemiology and Reporting Characteristics of Systematic Reviews of Biomedical Research: A Cross-Sectional Study
- PMID: 27218655
- PMCID: PMC4878797
- DOI: 10.1371/journal.pmed.1002028
Epidemiology and Reporting Characteristics of Systematic Reviews of Biomedical Research: A Cross-Sectional Study
Abstract
Background: Systematic reviews (SRs) can help decision makers interpret the deluge of published biomedical literature. However, a SR may be of limited use if the methods used to conduct the SR are flawed, and reporting of the SR is incomplete. To our knowledge, since 2004 there has been no cross-sectional study of the prevalence, focus, and completeness of reporting of SRs across different specialties. Therefore, the aim of our study was to investigate the epidemiological and reporting characteristics of a more recent cross-section of SRs.
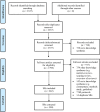
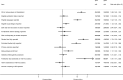
Methods and findings: We searched MEDLINE to identify potentially eligible SRs indexed during the month of February 2014. Citations were screened using prespecified eligibility criteria. Epidemiological and reporting characteristics of a random sample of 300 SRs were extracted by one reviewer, with a 10% sample extracted in duplicate. We compared characteristics of Cochrane versus non-Cochrane reviews, and the 2014 sample of SRs versus a 2004 sample of SRs. We identified 682 SRs, suggesting that more than 8,000 SRs are being indexed in MEDLINE annually, corresponding to a 3-fold increase over the last decade. The majority of SRs addressed a therapeutic question and were conducted by authors based in China, the UK, or the US; they included a median of 15 studies involving 2,072 participants. Meta-analysis was performed in 63% of SRs, mostly using standard pairwise methods. Study risk of bias/quality assessment was performed in 70% of SRs but was rarely incorporated into the analysis (16%). Few SRs (7%) searched sources of unpublished data, and the risk of publication bias was considered in less than half of SRs. Reporting quality was highly variable; at least a third of SRs did not report use of a SR protocol, eligibility criteria relating to publication status, years of coverage of the search, a full Boolean search logic for at least one database, methods for data extraction, methods for study risk of bias assessment, a primary outcome, an abstract conclusion that incorporated study limitations, or the funding source of the SR. Cochrane SRs, which accounted for 15% of the sample, had more complete reporting than all other types of SRs. Reporting has generally improved since 2004, but remains suboptimal for many characteristics.
Conclusions: An increasing number of SRs are being published, and many are poorly conducted and reported. Strategies are needed to help reduce this avoidable waste in research.
Conflict of interest statement
I have read the journal's policy and the authors of this manuscript have the following competing interests: MJP is a research fellow for the Australasian Cochrane Centre. ACT and FCL are authors of three of the systematic reviews included in this study, but were not involved in eligibility assessment or data extraction. DM is a member of
Figures


Similar articles
-
Folic acid supplementation and malaria susceptibility and severity among people taking antifolate antimalarial drugs in endemic areas.Cochrane Database Syst Rev. 2022 Feb 1;2(2022):CD014217. doi: 10.1002/14651858.CD014217. Cochrane Database Syst Rev. 2022. PMID: 36321557 Free PMC article.
-
Epidemiology and reporting characteristics of systematic reviews.PLoS Med. 2007 Mar 27;4(3):e78. doi: 10.1371/journal.pmed.0040078. PLoS Med. 2007. PMID: 17388659 Free PMC article.
-
An evaluation of epidemiological and reporting characteristics of complementary and alternative medicine (CAM) systematic reviews (SRs).PLoS One. 2013;8(1):e53536. doi: 10.1371/journal.pone.0053536. Epub 2013 Jan 14. PLoS One. 2013. PMID: 23341949 Free PMC article.
-
Epidemiology, quality, and reporting characteristics of systematic reviews and meta-analyses of nursing interventions published in Chinese journals.Nurs Outlook. 2015 Jul-Aug;63(4):446-455.e4. doi: 10.1016/j.outlook.2014.11.020. Epub 2014 Dec 4. Nurs Outlook. 2015. PMID: 26187084 Review.
-
Epidemiology, quality, and reporting characteristics of systematic reviews of acupuncture interventions published in Chinese journals.J Altern Complement Med. 2012 Sep;18(9):813-7. doi: 10.1089/acm.2011.0274. J Altern Complement Med. 2012. PMID: 22924413 Review.
Cited by
-
A framework for prospective, adaptive meta-analysis (FAME) of aggregate data from randomised trials.PLoS Med. 2021 May 6;18(5):e1003629. doi: 10.1371/journal.pmed.1003629. eCollection 2021 May. PLoS Med. 2021. PMID: 33956789 Free PMC article.
-
The PRISMA 2020 statement: An updated guideline for reporting systematic reviews.PLoS Med. 2021 Mar 29;18(3):e1003583. doi: 10.1371/journal.pmed.1003583. eCollection 2021 Mar. PLoS Med. 2021. PMID: 33780438 Free PMC article.
-
Evidence-based health policy in Germany: lack of communication and coordination between academia and health authorities?Syst Rev. 2023 Mar 13;12(1):36. doi: 10.1186/s13643-023-02204-6. Syst Rev. 2023. PMID: 36907893 Free PMC article.
-
Reporting quality of abstracts of systematic reviews/meta-analyses: An appraisal of Arab Journal of Urology across 12 years: the PRISMA-Abstracts checklist.Arab J Urol. 2022 Aug 22;21(1):52-65. doi: 10.1080/2090598X.2022.2113127. eCollection 2023. Arab J Urol. 2022. PMID: 36818377 Free PMC article.
-
EMG biofeedback combined with rehabilitation training may be the best physical therapy for improving upper limb motor function and relieving pain in patients with the post-stroke shoulder-hand syndrome: A Bayesian network meta-analysis.Front Neurol. 2023 Jan 10;13:1056156. doi: 10.3389/fneur.2022.1056156. eCollection 2022. Front Neurol. 2023. PMID: 36703623 Free PMC article.
References
-
- US National Library of Medicine. Key MEDLINE indicators. 2015 [cited 1 Sep 2015]. Available: http://www.nlm.nih.gov/bsd/bsd_key.html.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

