Artemether for severe malaria
- PMID: 25209020
- PMCID: PMC4455227
- DOI: 10.1002/14651858.CD010678.pub2
Artemether for severe malaria
Update in
-
Artemether for severe malaria.Cochrane Database Syst Rev. 2019 Jun 18;6(6):CD010678. doi: 10.1002/14651858.CD010678.pub3. Cochrane Database Syst Rev. 2019. PMID: 31210357 Free PMC article.
Abstract
Background: In 2011 the World Health Organization (WHO) recommended parenteral artesunate in preference to quinine as first-line treatment for people with severe malaria. Prior to this recommendation, many countries, particularly in Africa, had begun to use artemether, an alternative artemisinin derivative. This review evaluates intramuscular artemether compared with both quinine and artesunate.
Objectives: To assess the efficacy and safety of intramuscular artemether versus any other parenteral medication in treating severe malaria in adults and children.
Search methods: We searched the Cochrane Infectious Diseases Group Specialized Register, CENTRAL (The Cochrane Library), MEDLINE, EMBASE and LILACS, ISI Web of Science, conference proceedings and reference lists of articles. We also searched the WHO clinical trial registry platform, ClinicalTrials.gov and the metaRegister of Controlled Trials (mRCT) for ongoing trials up to 9 April 2014.
Selection criteria: Randomized controlled trials (RCTs) comparing intramuscular artemether with intravenous or intramuscular antimalarial for treating severe malaria.
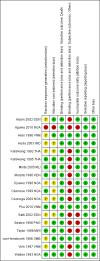
Data collection and analysis: The primary outcome was all-cause death.Two authors independently assessed trial eligibility, risk of bias and extracted data. We summarized dichotomous outcomes using risk ratios (RR) and continuous outcomes using mean differences (MD), and presented both measures with 95% confidence intervals (CI). Where appropriate, we combined data in meta-analyses and assessed the quality of the evidence using the GRADE approach.
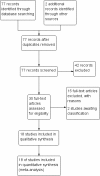
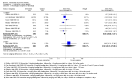
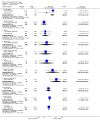
Main results: We included 18 RCTs, enrolling 2662 adults and children with severe malaria, carried out in Africa (11) and in Asia (7). Artemether versus quinine For children in Africa, there is probably little or no difference in the risk of death between intramuscular artemether and quinine (RR 0.96, 95% CI 0.76 to 1.20; 12 trials, 1447 participants, moderate quality evidence). Coma recovery may be about five hours shorter with artemether (MD -5.45, 95% CI -7.90 to -3.00; six trials, 358 participants, low quality evidence), and artemether may result in fewer neurological sequelae, but larger trials would be needed to confirm this (RR 0.84, 95% CI 0.66 to 1.07; seven trials, 968 participants, low quality evidence). Artemether probably shortens the parasite clearance time by about nine hours (MD -9.03, 95% CI -11.43 to -6.63; seven trials, 420 participants, moderate quality evidence), and may shorten the fever clearance time by about three hours (MD -3.73, 95% CI -6.55 to -0.92; eight trials, 457 participants, low quality evidence).For adults in Asia, treatment with intramuscular artemether probably results in fewer deaths than treatment with quinine (RR 0.59, 95% CI 0.42 to 0.83; four trials, 716 participants, moderate quality evidence). Artemether versus artesunate Artemether and artesunate have not been directly compared in randomized trials in African children.For adults in Asia, mortality is probably higher with intramuscular artemether (RR 1.80, 95% CI 1.09 to 2.97, two trials,494 participants, moderate quality evidence).
Authors' conclusions: Although there is a lack of direct evidence comparing artemether with artesunate, artemether is probably less effective than artesunate at preventing deaths from severe malaria. In circumstances where artesunate is not available, artemether is an alternative to quinine.
Conflict of interest statement
None known.
Figures












Similar articles
-
Artemether for severe malaria.Cochrane Database Syst Rev. 2019 Jun 18;6(6):CD010678. doi: 10.1002/14651858.CD010678.pub3. Cochrane Database Syst Rev. 2019. PMID: 31210357 Free PMC article.
-
Artesunate versus quinine for treating severe malaria.Cochrane Database Syst Rev. 2007 Oct 17;(4):CD005967. doi: 10.1002/14651858.CD005967.pub2. Cochrane Database Syst Rev. 2007. Update in: Cochrane Database Syst Rev. 2011 Mar 16;(3):CD005967. doi: 10.1002/14651858.CD005967.pub3. PMID: 17943870 Updated. Review.
-
Artesunate plus pyronaridine for treating uncomplicated Plasmodium falciparum malaria.Cochrane Database Syst Rev. 2014 Mar 4;(3):CD006404. doi: 10.1002/14651858.CD006404.pub2. Cochrane Database Syst Rev. 2014. Update in: Cochrane Database Syst Rev. 2019 Jan 08;1:CD006404. doi: 10.1002/14651858.CD006404.pub3. PMID: 24596021 Free PMC article. Updated. Review.
-
Artesunate versus quinine for treating severe malaria.Cochrane Database Syst Rev. 2011 Mar 16;(3):CD005967. doi: 10.1002/14651858.CD005967.pub3. Cochrane Database Syst Rev. 2011. Update in: Cochrane Database Syst Rev. 2012 Jun 13;(6):CD005967. doi: 10.1002/14651858.CD005967.pub4. PMID: 21412892 Updated. Review.
-
Artesunate versus quinine for treating severe malaria.Cochrane Database Syst Rev. 2012 Jun 13;2012(6):CD005967. doi: 10.1002/14651858.CD005967.pub4. Cochrane Database Syst Rev. 2012. PMID: 22696354 Free PMC article. Review.
Cited by
-
Effects of artemether on the proliferation, apoptosis, and differentiation of keratinocytes: potential application for psoriasis treatment.Int J Clin Exp Med. 2015 May 15;8(5):7069-78. eCollection 2015. Int J Clin Exp Med. 2015. PMID: 26221244 Free PMC article.
-
Artemether for severe malaria.Cochrane Database Syst Rev. 2019 Jun 18;6(6):CD010678. doi: 10.1002/14651858.CD010678.pub3. Cochrane Database Syst Rev. 2019. PMID: 31210357 Free PMC article.
-
Targeted Therapy of B7 Family Checkpoints as an Innovative Approach to Overcome Cancer Therapy Resistance: A Review from Chemotherapy to Immunotherapy.Molecules. 2022 May 31;27(11):3545. doi: 10.3390/molecules27113545. Molecules. 2022. PMID: 35684481 Free PMC article. Review.
-
The growing pipeline of natural aminoacyl-tRNA synthetase inhibitors for malaria treatment.Bioengineered. 2016 Apr 2;7(2):60-4. doi: 10.1080/21655979.2016.1149270. Epub 2016 Mar 10. Bioengineered. 2016. PMID: 26963157 Free PMC article.
-
Artemether Regulates Chemosensitivity to Doxorubicin via Regulation of B7-H3 in Human Neuroblastoma Cells.Med Sci Monit. 2017 Sep 3;23:4252-4259. doi: 10.12659/msm.902068. Med Sci Monit. 2017. PMID: 28866709 Free PMC article.
References
References to studies included in this review
-
- Adam I, Idris HM, Mohamed‐Ali AA, Aelbasit, Elbashir MI. Comparison of intramuscular artemether and intravenous quinine in the treatment of Sudanese children with severe falciparum malaria. East African Medical Journal 2002;79(12):621‐5. - PubMed
-
- Aguwa CN, Ukwe CV, Adibe MO. A comparative study of quinine and artemether in the treatment of severe malaria in Nigerian children. Tropical Journal of Pharmaceutical Research 2010;9(1):11‐7.
-
- Tran TH, Day NP, Nguyen HP, Nguyen TH, Tran TH, Pham PL, et al. A controlled trial of artemether or quinine in Vietnamese adults with severe falciparum malaria. New England Journal of Medicine 1996;335(2):76‐83. - PubMed
-
- Huda SN, Shahab T, Ali SM, Afzal K, Khan HM. A comparative clinical trial of artemether and quinine in children with severe malaria. Indian Pediatrics 2003;40(10):939‐45. - PubMed
-
- Karbwang J, Sukontason K, Rimchala W, Namsiripongpun W, Tin T, Auprayoon P, et al. Preliminary report: a comparative clinical trial of artemether and quinine in severe falciparum malaria. Southeast Asian Journal of Tropical Medicine and Public Health 1992;23(4):768‐72. - PubMed
References to studies excluded from this review
-
- Bhattacharya PC, Pai‐dhungat AJ, Patel K. Artemether in moderate to severe malaria: a multicenter trial in India. Southeast Asian Journal of Tropical Medicine and Public Health 1997;28(4):736‐40. - PubMed
-
- Bunnag D, Karbwang J, Harinasuta T. Artemether in the treatment of multiple drug resistant falciparum malaria. Southeast Asian Journal of Tropical Medicine and Public Health 1992;23(4):762‐7. - PubMed
-
- Falade CO, Fadero FF, Happi CT, Dada‐Adegbola HO, Gbotosho GO, Ayede I, et al. Dihydroartemisinin suppository in moderately severe malaria: comparative efficacy of dihydroartemisinin suppository versus intramuscular artemether followed by oral sulfadoxine‐pyrimethamine in the management of moderately severe malaria in Nigerian children. American Journal of Tropical Medicine and Hygiene 2007;76(1):1‐6. - PubMed
References to studies awaiting assessment
-
- Danis M, Chandenier J, Doumbo O, Kombila M, Kouame J, Louis F, et al. Results obtained with i.m. artemether versus i.v. quinine in the treatment of severe malaria in a multi‐centre study in Africa. Japan Journal of Tropical Medicine and Hygiene 1996;24(Suppl 1):93‐6.
-
- Faiz MA, Rahman E, Hossain MA, Rahman MR, Yunus EB, Samad R, et al. A randomized controlled trial comparing artemether and quinine in the treatment of cerebral malaria in Bangladesh. Indian Journal of Malariology 2001;38(1‐2):9‐18. - PubMed
Additional references
-
- Artemether‐Quinine Meta‐analysis Study Group. A meta‐analysis using individual patient data of trials comparing artemether with quinine in the treatment of severe falciparum malaria. Transactions of the Royal Society of Tropical Medicine and Hygiene 2001;95(6):637‐50. - PubMed
-
- Dondorp A, Nosten F, Stepniewska K, Day N, White N, South East Asian Quinine Artesunate Malaria Trial (SEAQUAMAT) group. Artesunate versus quinine for treatment of severe falciparum malaria: a randomised trial. Lancet 2005;366(9487):717‐25. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

