Newborn screening for galactosemia in the United States: looking back, looking around, and looking ahead
- PMID: 24718839
- PMCID: PMC4413015
- DOI: 10.1007/8904_2014_302
Newborn screening for galactosemia in the United States: looking back, looking around, and looking ahead
Erratum in
-
Erratum to: Newborn Screening for Galactosemia in the United States: Looking Back, Looking Around, and Looking Ahead.JIMD Rep. 2015;15:133. doi: 10.1007/8904_2014_369. Epub 2014 Nov 8. JIMD Rep. 2015. PMID: 25381176 Free PMC article. No abstract available.
Abstract
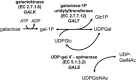
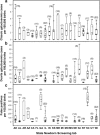
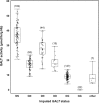
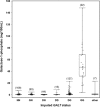
It has been 50 years since the first newborn screening (NBS) test for galactosemia was conducted in Oregon, and almost 10 years since the last US state added galactosemia to their NBS panel. During that time an estimated >2,500 babies with classic galactosemia have been identified by NBS. Most of these infants were spared the trauma of acute disease by early diagnosis and intervention, and many are alive today because of NBS. Newborn screening for galactosemia is a success story, but not yet a story with a completely happy ending. NBS, follow-up testing, and intervention for galactosemia continue to present challenges that highlight gaps in our knowledge. Here we compare galactosemia screening and follow-up data from 39 NBS programs gathered from the states directly or from public sources. On some matters the programs agreed: for example, those providing relevant data all identify classic galactosemia in close to 1/50,000 newborns and recommend immediate and lifelong dietary restriction of galactose for those infants. On other matters the programs disagree. For example, Duarte galactosemia (DG) detection rates vary dramatically among states, largely reflecting differences in screening approach. For infants diagnosed with DG, >80% of the programs surveyed recommend complete or partial dietary galactose restriction for the first year of life, or give mixed recommendations; <20% recommend no intervention. This disparity presents an ongoing dilemma for families and healthcare providers that could and should be resolved.
Figures
Similar articles
-
Galactosemia: when is it a newborn screening emergency?Mol Genet Metab. 2012 May;106(1):7-11. doi: 10.1016/j.ymgme.2012.03.007. Epub 2012 Mar 21. Mol Genet Metab. 2012. PMID: 22483615 Review.
-
Developmental Outcomes of School-Age Children with Duarte Galactosemia: A Pilot Study.JIMD Rep. 2015;19:75-84. doi: 10.1007/8904_2014_370. Epub 2015 Feb 15. JIMD Rep. 2015. PMID: 25681083 Free PMC article.
-
Nine years of newborn screening for classical galactosemia in the Netherlands: Effectiveness of screening methods, and identification of patients with previously unreported phenotypes.Mol Genet Metab. 2017 Mar;120(3):223-228. doi: 10.1016/j.ymgme.2016.12.012. Epub 2016 Dec 29. Mol Genet Metab. 2017. PMID: 28065439
-
Diversity of approaches to classic galactosemia around the world: a comparison of diagnosis, intervention, and outcomes.J Inherit Metab Dis. 2012 Nov;35(6):1037-49. doi: 10.1007/s10545-012-9477-y. Epub 2012 Mar 27. J Inherit Metab Dis. 2012. PMID: 22450714 Free PMC article.
-
Drosophila melanogaster Models of Galactosemia.Curr Top Dev Biol. 2017;121:377-395. doi: 10.1016/bs.ctdb.2016.07.009. Epub 2016 Aug 3. Curr Top Dev Biol. 2017. PMID: 28057307 Free PMC article. Review.
Cited by
-
The Importance of Neonatal Screening for Galactosemia.Nutrients. 2022 Dec 20;15(1):10. doi: 10.3390/nu15010010. Nutrients. 2022. PMID: 36615667 Free PMC article. Review.
-
Long-term complications in classic galactosemia are not progressive.Mol Genet Metab. 2023 Nov;140(3):107708. doi: 10.1016/j.ymgme.2023.107708. Epub 2023 Oct 11. Mol Genet Metab. 2023. PMID: 37866059 Free PMC article.
-
Genomic Sequencing for Newborn Screening: Results of the NC NEXUS Project.Am J Hum Genet. 2020 Oct 1;107(4):596-611. doi: 10.1016/j.ajhg.2020.08.001. Epub 2020 Aug 26. Am J Hum Genet. 2020. PMID: 32853555 Free PMC article.
-
Establishing New Cut-Off Limits for Galactose 1-Phosphate-Uridyltransferase Deficiency for the Dutch Newborn Screening Programme.JIMD Rep. 2017;32:1-6. doi: 10.1007/8904_2016_560. Epub 2016 May 21. JIMD Rep. 2017. PMID: 27207469 Free PMC article.
-
The risk of classical galactosaemia in newborns with borderline galactose metabolites on newborn screening.JIMD Rep. 2022 Sep 21;64(2):180-186. doi: 10.1002/jmd2.12339. eCollection 2023 Mar. JIMD Rep. 2022. PMID: 36873086 Free PMC article.
References
-
- Alano A, Almashanu S, Maceratesi P, Reichardt J, Panny S, Cowan TM. UDP-galactose-4-epimerase deficiency among African-Americans: evidence for multiple alleles. J Investig Med. 1997;45:191A.
-
- Beutler E, Baluda M. A simple spot screening test for galactosemia. J Lab Clin Med. 1966;68:137–141. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

